What Is a Dialysis Access?
Dialysis needs a way to reach your blood. Your access is the connection that makes this possible — every session, blood leaves through it, passes through the dialysis machine, and returns cleaned. There are four main types.
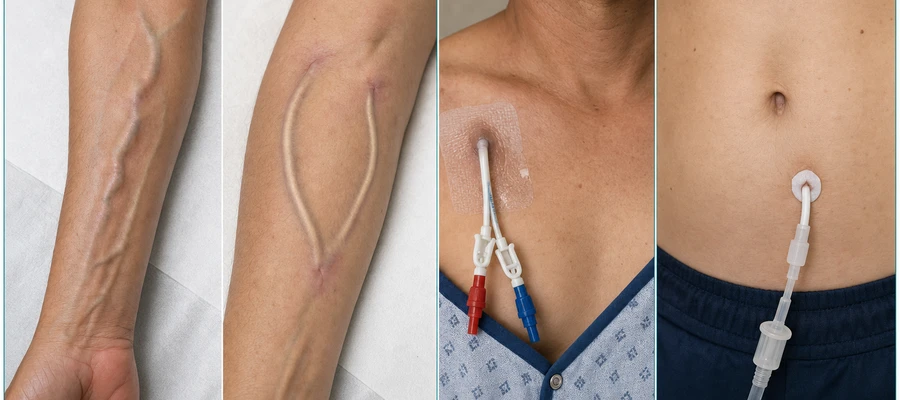
AV Fistula
A surgeon connects an artery and a vein in your arm, usually at the wrist or elbow. Blood flowing through this connection creates a buzzing sensation called the "thrill." It is the best long-term access and takes 6–8 weeks to mature before it can be used. It has the lowest risk of infection of all access types.
AV Graft
A soft plastic tube connects an artery and a vein in the arm. It is ready for use sooner than a fistula (2–4 weeks) but carries a slightly higher infection risk because the plastic material can harbor bacteria more easily than your own blood vessel.
Tunneled Catheter (Permcath)
A two-lumen tube is placed under the skin of the chest and into a large vein near the heart. It is used when a fistula or graft is not yet ready or not possible. It carries the highest infection risk of all access types because it provides a direct path from the skin surface to the bloodstream.
PD Catheter
A soft silicone tube passes through the abdominal wall into the belly cavity (peritoneal space). It is used for peritoneal dialysis, which is often done at home. Infection at the exit site, along the tunnel, or inside the belly (peritonitis) is the most serious complication of PD.
Why infection is an emergency
Bacteria entering through a dialysis access go directly into your bloodstream during every dialysis session. What starts as a local redness can become a life-threatening bloodstream infection (sepsis) or endocarditis within hours. Do not wait to see if it gets better on its own.
Daily Care
Most access infections begin with small breaks in care. The habits below are your strongest defense.
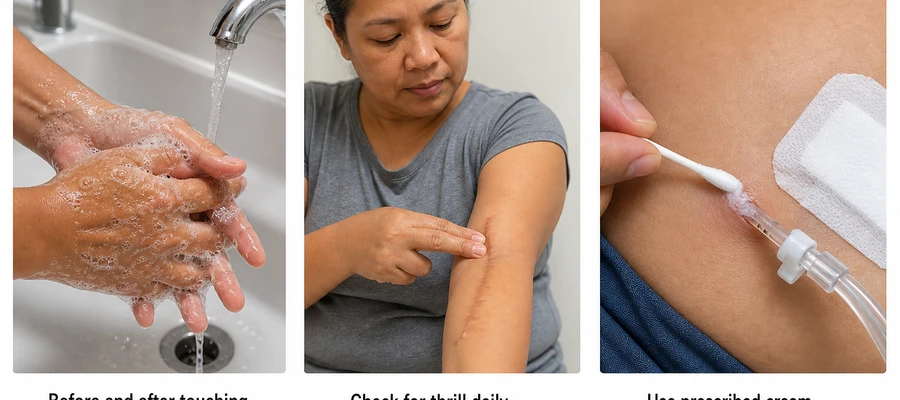
Fistula and Graft
- Inspect your access every morning — run your finger gently along it. You should feel the thrill (a buzzing or vibration). If the thrill is gone, call your dialysis center immediately.
- Wash the access site with soap and water daily.
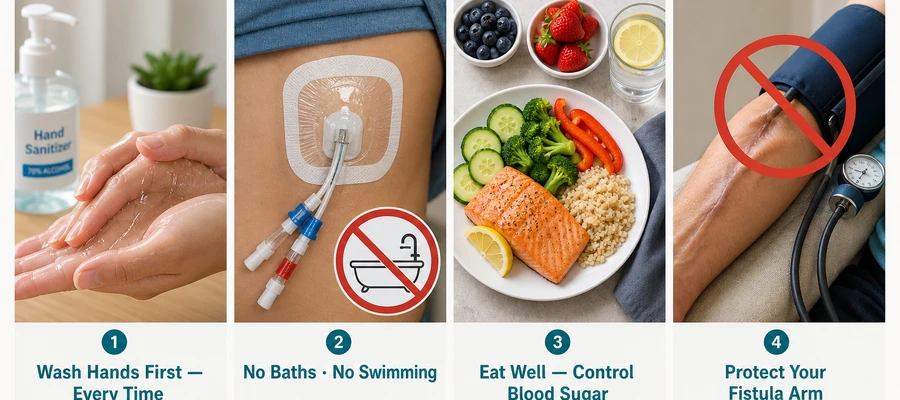
- Never allow blood pressure cuffs, blood draws, or IV lines on your access arm — this is an absolute rule, no exceptions.
- Do not wear tight clothing, wristwatches, or jewelry over or near the access site.
- Avoid sleeping directly on the access arm — this can compress blood flow and increase clot and infection risk.
- Keep fingernails short and avoid scratching over needle puncture sites.
- After each dialysis session: check that needle sites have stopped bleeding completely before leaving the center. Apply a gentle clean dressing if advised by your nurse.
Tunneled Catheter (Permcath)
- Keep the catheter exit site covered with a clean, dry dressing at all times between sessions.
- Do not shower without a proper waterproof cover applied over the site — ask your dialysis nurse which cover to use and how to apply it.
- Never submerge the catheter site in baths, swimming pools, or the sea — ever.
- Never disconnect or reconnect the catheter caps yourself unless you have been formally trained and specifically instructed by your medical team.
- Report to your center immediately if the dressing falls off, the catheter appears to have shifted position, or you see any redness or discharge at the exit site.
- Dressing changes are done by trained dialysis staff only — do not attempt this at home.
- If your catheter has clamps, never leave them open (unclamped) when not in use.
PD Catheter (Exit Site)
- Clean your exit site daily using soap and water (or chlorhexidine if your doctor prescribed it). Always wash your hands thoroughly with soap and water first.
- After cleaning, dry the site completely with clean gauze — moisture under the dressing is a key infection trigger.
- Apply mupirocin or gentamicin cream (if prescribed) directly to the exit site with a clean cotton swab after drying.
- Secure your catheter with tape so it does not move or pull — the catheter moving in and out of the tunnel is one of the most important risk factors for infection.
- Wear loose clothing that does not press on or rub against the catheter.
- Brief showering is acceptable, but never take baths or swim in pools or open water.
- Never pull or rotate the catheter unless your nurse specifically instructed you to do so.
Prevention Best Practices
Hand Hygiene — Most Important
Hand Hygiene Checklist
- Wash hands with soap and water for at least 20 seconds before and after touching any part of your access or its dressing.
- Use alcohol-based hand sanitizer if soap is not immediately available — but soap and water is better when your hands are visibly dirty.
- You have the right to ask any dialysis staff member to wash or sanitize their hands in front of you before touching your access. This is your right and your safety.
Showering and Water Exposure
- Fistula/graft: Showering is fine. Avoid submerging the access arm in baths or pools. No open-water swimming until needle puncture sites are fully healed.
- Tunneled catheter: No showering without waterproof protection properly applied. No baths, pools, or sea — ever, without exception.
- PD catheter: Brief showering is acceptable. No baths or swimming pools.
Mupirocin / Gentamicin Exit-Site Cream
- If your doctor prescribed exit-site cream, apply it every day after cleaning and drying the site — do not skip even a single day.
- This is one of the strongest proven steps for preventing PD exit-site and tunnel infections.
- Apply a small amount with a clean cotton swab; do not use your fingers directly on the site.
Nutrition and Blood Sugar
- Patients who are malnourished or who have poorly controlled diabetes develop access infections at much higher rates — your immune system depends on good nutrition.
- Aim for adequate protein intake. Your dietitian will advise specific targets based on your dialysis type.
- Keep blood sugar as well-controlled as possible — high glucose directly impairs your immune response and wound healing.
Other Important Prevention Points
- Tell your dialysis team immediately about any skin breaks, boils, pimples, cuts, or dental procedures — even bacteria from dental work can travel in the blood and seed your access.
- If your doctor prescribed mupirocin nasal ointment for MRSA, apply it exactly as instructed (usually 3 times daily for 5 days, repeated monthly).
- Never allow anyone to access your catheter or PD port outside your dialysis center without proper training and sterile equipment.
- Avoid picking at or scratching needle puncture sites after dialysis — even a small scratch can introduce bacteria.
Warning Signs — When to Act
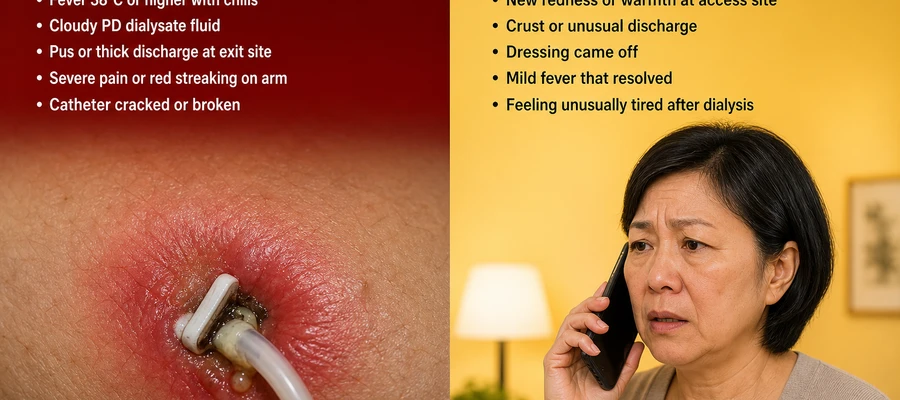
Go to the Emergency Room immediately if you have ANY of these:
- Fever at or above 38°C (100.4°F) — especially with chills or shaking
- Fever with confusion, extreme weakness, or a very fast heartbeat — this may be sepsis
- Pus or thick discharge coming from your catheter exit site or PD exit site
- Severe pain, swelling, or red streaking spreading up the arm from your fistula or graft
- Your catheter cracked, broke, or came apart at any point
- Your PD dialysis fluid (dialysate) appears cloudy — this is peritonitis until proven otherwise
Call your dialysis clinic today — do not wait until your next scheduled session:
- New redness, warmth, or mild swelling around fistula needle sites or any exit site
- Crust or dried discharge at the PD exit site that was not there before
- Dressing fell off and the site looks different from how it normally appears
- A pimple or boil developing anywhere near the access site
- You had a fever that went away on its own
- You feel generally unwell or unusually tired in the days after dialysis
When in doubt — call
Infections caught early are treated with antibiotics. Infections caught late may require surgery, catheter removal, or weeks in hospital. A single phone call could prevent all of that.
Be prepared
Save your dialysis center's phone number in your mobile phone right now. Know the location of the nearest emergency room that has dialysis capability.
At Your Dialysis Center
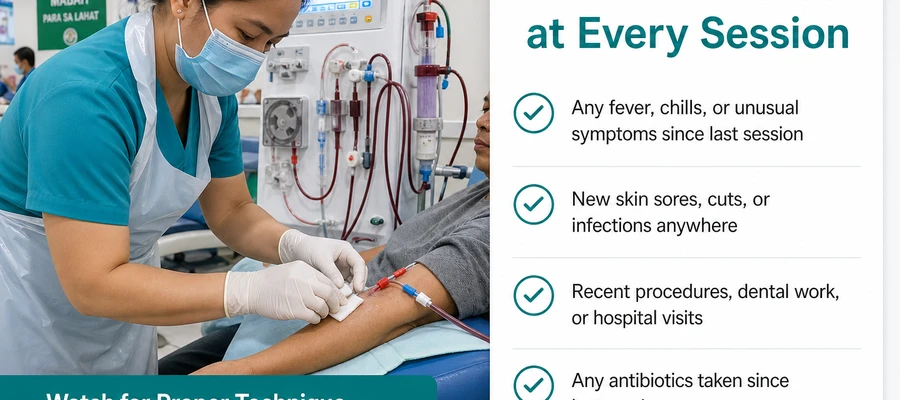
What Good Technique Looks Like
Every time staff connect you to dialysis, they should wash or sanitize their hands, put on gloves, and clean your access site with an antiseptic solution before touching the needles or catheter. This is a standard requirement — not an optional courtesy.
If you observe a break in technique — for example, a staff member touches something non-sterile and then goes to connect you without re-gloving — you have the right to politely but firmly say: "Could you please change your gloves before connecting me?" Your safety depends on it.
What to Tell the Nurse at the Start of Every Session
- Any fever, chills, or unusual symptoms since your last dialysis session
- Any new skin sores, cuts, wounds, or infections anywhere on your body
- Any recent dental procedures, medical procedures, or hospital visits
- Any antibiotic use since your last session
Buttonhole Cannulation (Fistula)
Some dialysis centers use a "buttonhole" technique where the same needle path is used at each session. If your center uses this method, the nurse must remove the scab from the track cleanly before inserting the needle. If you see blood or pus in the track, alert your nurse before the needle goes in. Never try to remove the scab yourself at home.

W. G. M. Rivero, MD, FPCP, DPSN
Nephrology and Internal Medicine specialist. This guide is written to help patients and families recognize access infections early, take the right steps, and work confidently with their dialysis team. When in doubt, always reach out to your healthcare provider.
Diplomate, Philippine Society of Nephrology · Fellow, Philippine College of Physicians
Common Questions
My exit site is slightly pink — is that normal?
Pink skin without any discharge, crust, or pain can be normal immediately after cleaning, especially when the site is new. However, if the pinkness persists for more than a day or two, spreads, or is accompanied by any crust, pus, or swelling, call your dialysis team. Do not dismiss it.
Can I shower with my Permcath?
Only if a waterproof dressing cover is properly applied and secured before you shower. The cover must protect the exit site completely. When in doubt, take a sponge bath around the site instead. Never submerge the catheter in any water.
I have a fever but my access looks completely normal — should I still worry?
Yes. Catheter-related infections very often present with fever alone before any redness or discharge appears at the exit site. Fever in a dialysis patient with any type of catheter must be taken seriously. Call your dialysis center or go to the emergency room — do not wait.
My PD fluid looks a bit cloudy — can I wait until tomorrow's session to mention it?
No. Cloudy PD dialysate means peritonitis until proven otherwise. Go to your dialysis center or the emergency room now. Peritonitis that is not treated the same day can become life-threatening and may result in permanent loss of PD as a treatment option.
Can I use my access arm for blood pressure checks at a pharmacy?
No. Never put a blood pressure cuff on your fistula or graft arm — at any location, for any reason. Always inform the person checking your blood pressure and remind them to use the other arm.
The dialysis nurse did not wash her hands before connecting me — what should I do?
Politely say: "Excuse me, could you please wash or sanitize your hands before connecting me?" This is your right as a patient and a standard infection-control requirement. A professional nurse will understand and comply without offense.