Clinicians rarely struggle with what a drug does. They struggle with when to start it, what to check first, how fast to escalate, what a creatinine bump really means, and when to hold vs. stop. Each section below gives you that operational layer — directly actionable, with explicit thresholds and decision branches.
SGLT2 Inhibitors — Safe Start Protocol
SGLT2i are now standard of care for CKD (eGFR ≥20) and heart failure regardless of diabetes. The barriers to starting are almost always about fear of the initial creatinine dip and sick-day rules — both of which are manageable.
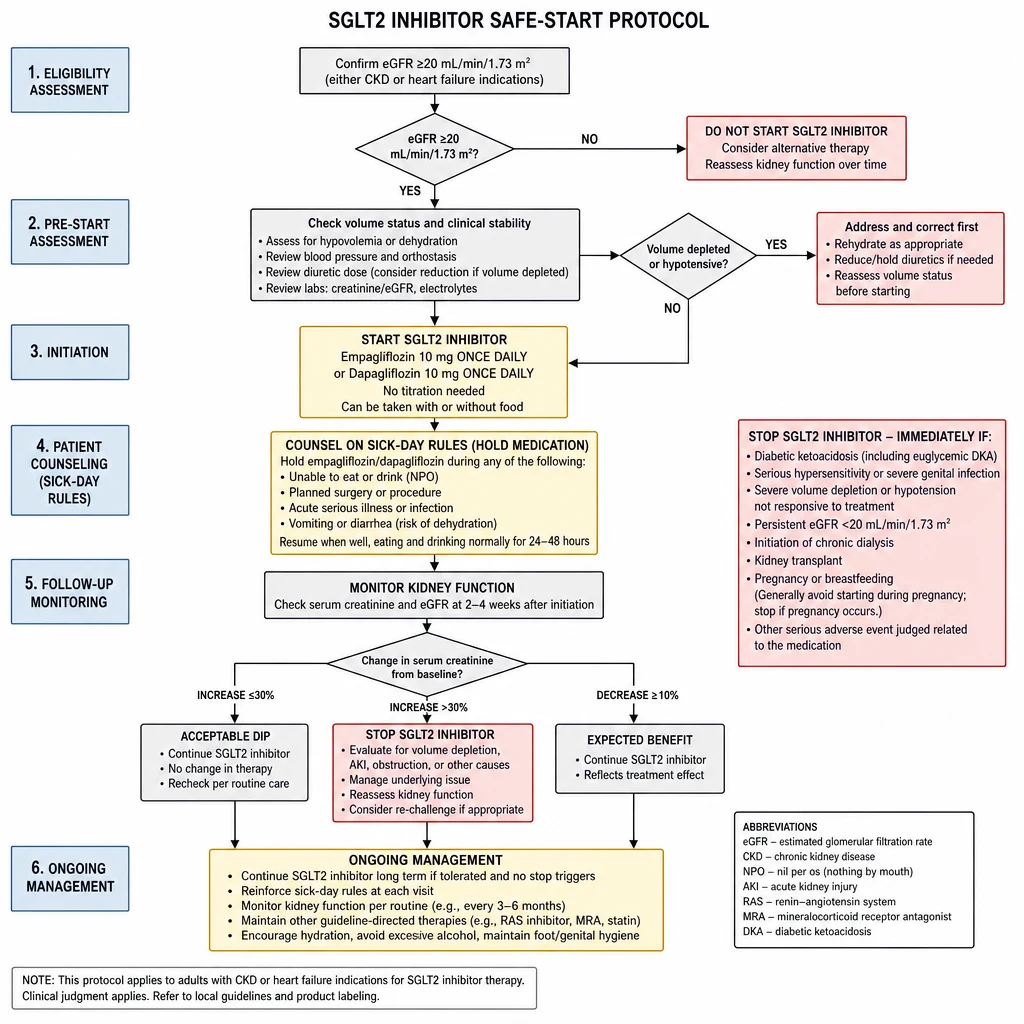 SGLT2i Safe-Start Protocol — eGFR thresholds, sick-day rules, and monitoring schedule.
SGLT2i Safe-Start Protocol — eGFR thresholds, sick-day rules, and monitoring schedule.
Glycemic benefit only: eGFR ≥25–45 depending on agent; check product PI.
Empagliflozin and dapagliflozin retain renoprotective efficacy down to eGFR 20.
Review concurrent insulin/sulfonylurea — may need dose reduction at initiation (hypoglycemia risk).
Note: eGFR will typically dip 3–5 mL/min in first 2–4 weeks. This is hemodynamic (efferent arteriolar relaxation), not structural injury.
Take any time, with or without food. Canagliflozin: give before first meal of day.
Presentation: nausea, vomiting, abdominal pain, fatigue, ketonemia — blood glucose often 100–200 mg/dL (not dramatically elevated).
Action: if suspected, hold SGLT2i, check urine/serum ketones and blood gas, refer to ER.
Fournier's gangrene (necrotizing fasciitis of perineum): very rare (<1:1,000,000) but serious. Instruct to report any perineal pain, swelling, or erythema immediately.
Canagliflozin: small increased risk of lower-limb amputation in high-risk PAD patients — assess vascular status first.
| Timepoint | What to check | Expected / Action |
|---|---|---|
| 2–4 weeks | eGFR, Creatinine, K⁺, BP | eGFR dip ≤30% = acceptable; stabilizes. Do NOT stop for this dip. |
| 3 months | eGFR, HbA1c (if T2D), weight | eGFR should stabilize or improve vs. pre-dip baseline. |
| Every 6 months | eGFR, K⁺, urinalysis | Ongoing renoprotective monitoring. |
| Any illness | eGFR, Cr | Hold drug; recheck before resuming. |
Quick Reference — SGLT2i at a Glance
- Start at eGFR ≥20; below this, discuss with nephrology
- Expected Cr dip at 2–4 weeks:
≤30%= acceptable, do not stop - Sick-day rule: HALT for NPO / surgery / infection / vomiting >24 h
- DKA can occur with near-normal glucose — check ketones if symptomatic
- Counsel on genital hygiene at every initiation visit
- Reduce insulin/SU dose at start if T2D — hypoglycemia risk
- Canagliflozin: assess PAD / amputation risk before starting
- Resume after illness/procedure: eating normally + 48 h post-procedure
ACEi / ARB — Creatinine Rise Interpretation
The single most common reason patients are inappropriately taken off ACEi/ARB is a creatinine bump that is both expected and protective. The key question is: is the rise hemodynamic or structural?
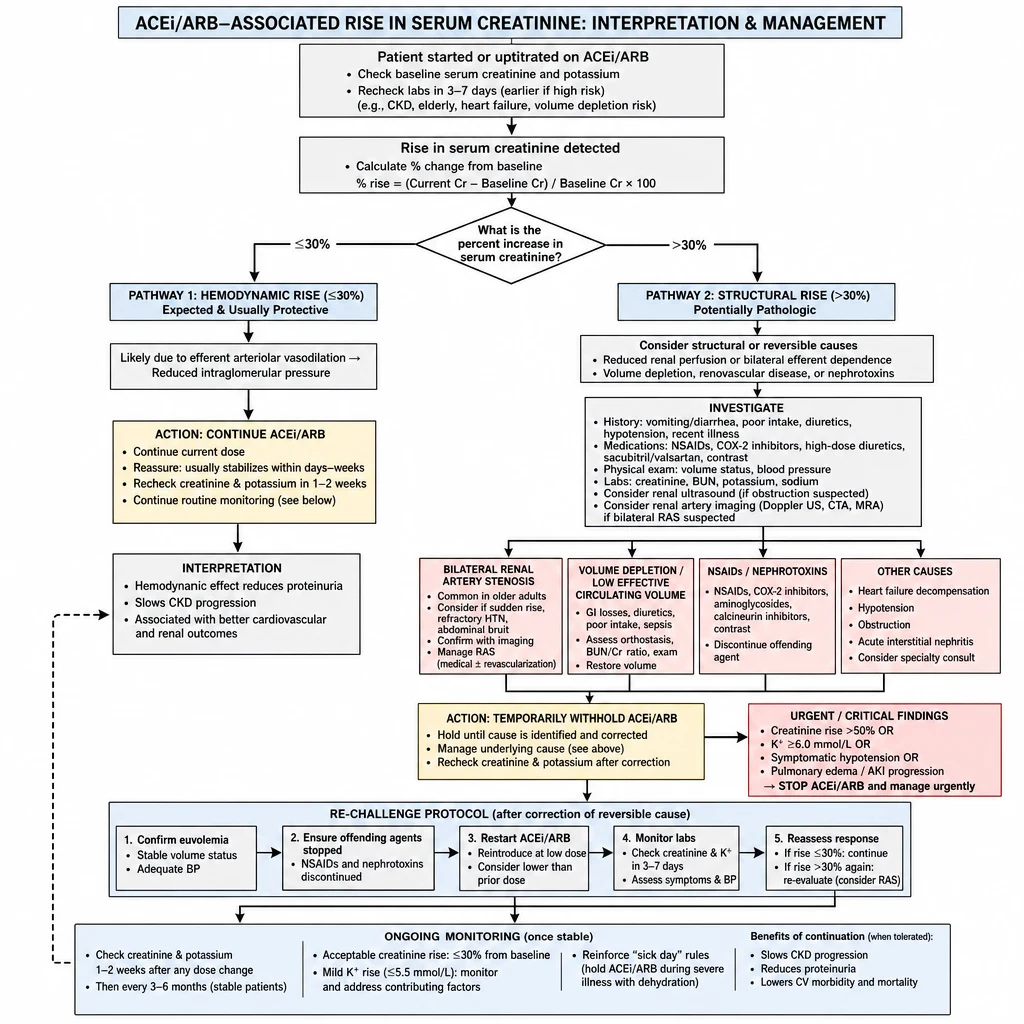 ACEi/ARB Creatinine Rise — hemodynamic (≤30%, continue) vs structural (>30%, investigate).
ACEi/ARB Creatinine Rise — hemodynamic (≤30%, continue) vs structural (>30%, investigate).
The Two-Mechanism Framework
Hemodynamic Rise — Expected & Protective
Mechanism: RAASi reduces efferent arteriolar constriction → lower intraglomerular pressure → transient drop in filtered creatinine.
Rise: ≤30% from baseline within 4 weeks.
Stabilizes by: 4–8 weeks.
This is the anti-proteinuric mechanism. Do NOT stop for this rise — doing so removes the protection you prescribed the drug for.
Structural Rise — Investigate & Act
Mechanism: Perfusion failure (bilateral RAS, severe volume depletion) or intrinsic renal injury.
Rise: >30% from baseline within 4 weeks, or rapidly progressive.
Does NOT stabilize — continues to worsen.
Identify and correct the reversible cause before restarting; hold drug if rise is severe or progressive.
| Rise from baseline | Classification | Action |
|---|---|---|
| <15% | Acceptable hemodynamic effect | Continue; recheck in 4 weeks |
| 15–30% | Acceptable; monitor closely | Continue; recheck in 2 weeks; check volume status |
| 30–50% | Investigate — likely reversible cause | Identify and correct reversible cause; may continue if correctable |
| >50% or >1 mg/dL above baseline | Reduce or hold; investigate bilateral RAS | Hold drug; evaluate for structural cause; restart only after resolution |
If suspected: hold ACEi/ARB, refer to nephrology/vascular, order renal artery duplex or CTA.
2. Wait until Cr returns to within 15% of pre-treatment baseline.
3. Restart at half the previous dose.
4. Recheck Cr in 1–2 weeks.
5. If rise <30% again, continue and uptitrate slowly.
The proteinuric patient with a Cr dip — do not stop
If a CKD patient on RAASi has a rise in Cr of 20% but proteinuria improves from 1.5 g/day to 0.6 g/day — this is a success, not a failure. The creatinine rise reflects reduced hyperfiltration. Stopping the drug eliminates the anti-proteinuric effect and accelerates CKD progression.
Quick Reference — ACEi/ARB Creatinine Rise
- Acceptable rise:
≤30%from baseline within 4 weeks — do not stop - Always check: volume status, NSAID use, bilateral RAS risk factors
- Bilateral RAS: asymmetric kidneys + resistant HTN + Cr rise = image renal arteries before restarting
- Re-challenge: fix reversible cause → restart at half dose → recheck in 1–2 weeks
- Avoid: ACEi + ARB combination (ONTARGET); combine with SGLT2i or finerenone instead
RAASi + Hyperkalemia — Mitigation Without Stopping
Stopping RAASi for hyperkalemia trades a short-term fix for long-term harm. The PRIORITIZE-HK trial showed this clearly: patients who stopped had higher mortality and more CV events. The goal is to manage the potassium, not remove the RAASi.
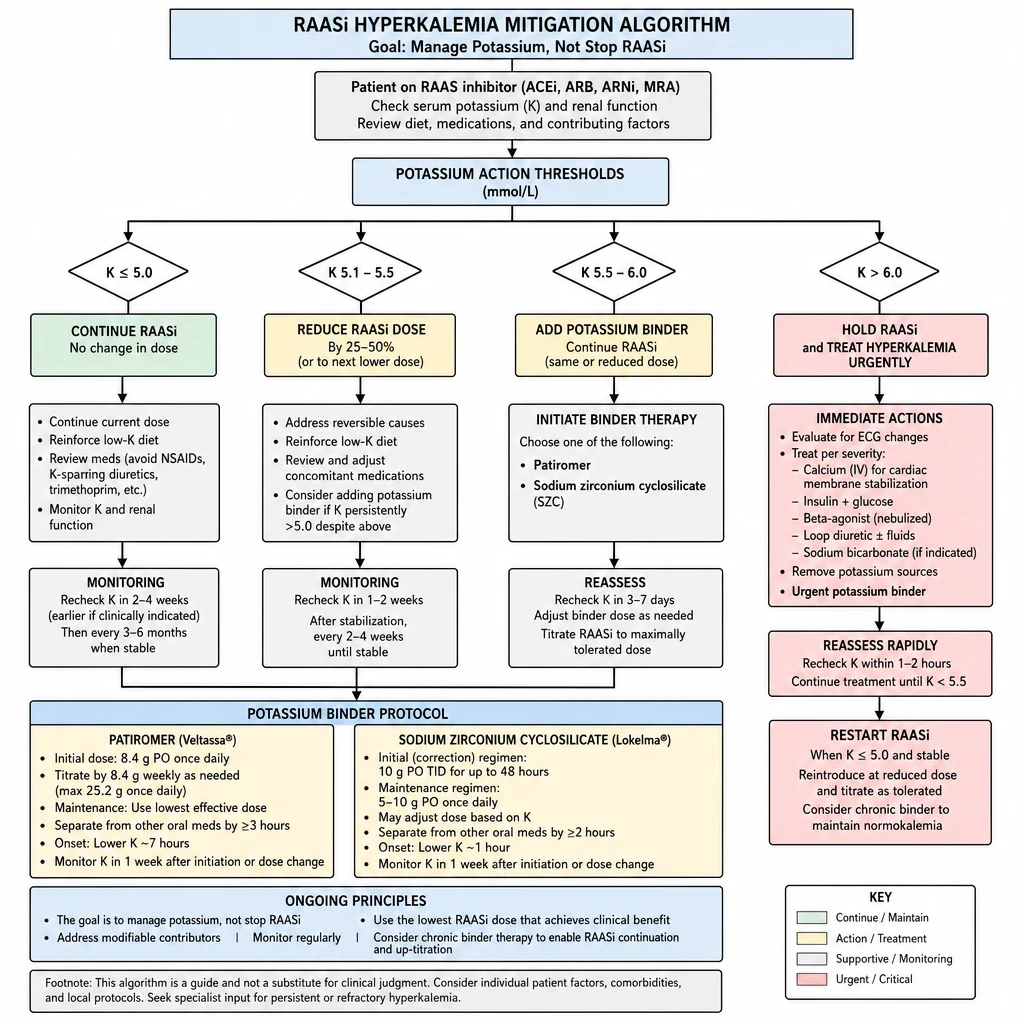 RAASi + Hyperkalemia — potassium thresholds, binder protocol, and dose-reduction ladder.
RAASi + Hyperkalemia — potassium thresholds, binder protocol, and dose-reduction ladder.
Potassium Monitoring — Action Thresholds
Potassium Binder Operational Guide
Kalimate (Calcium Polystyrene Sulfonate)
Brand: Kalimate (Kowa/MedChoice) · ~₱30–50/sachet (5 g)
Dose: 5–15 g QD–TID in water or juice (do not mix with fruit juice high in K⁺).
Onset: several hours — not for acute emergency management.
Advantage over SPS: exchanges Ca²⁺ for K⁺ (not Na⁺) — preferred in CHF, hypertension, and volume-sensitive patients; minimal sodium loading.
SE: constipation (most common — add lactulose if needed); GI upset; hypercalcemia with prolonged high-dose use (monitor Ca²⁺). Avoid in bowel obstruction or ileus.
Timing: give 2–3 h away from other oral medications to reduce adsorption interference.
Sodium Polystyrene Sulfonate (Kayexalate / Resonium A)
Dose: 15–60 g/day in divided doses orally or as retention enema (30–60 g in 150 mL water, retain 30–60 min).
Onset: 1–2 h (enema) to several hours (oral) — enema route acceptable for subacute management.
Caution — sodium load: each gram releases ~3 mEq Na⁺ — avoid or use with caution in CHF, severe HTN, and anasarca. Prefer Kalimate in fluid-overloaded patients.
SE: constipation; GI necrosis (rare — avoid in postoperative patients, ileus, or sorbitol co-administration); hypokalemia with overuse.
Note: always use sorbitol-free formulations; sorbitol co-administration linked to intestinal necrosis.
NSAIDs — reduce aldosterone effect + reduce GFR; always a target for removal
Spironolactone / eplerenone — essential in CHF; reduce dose or hold if K⁺ >5.5
Calcineurin inhibitors — tacrolimus > cyclosporine; check levels if hyperkalemia worsens
Beta-blockers — mild K⁺-retaining effect; generally continue
Heparin (high-dose) — impairs aldosterone synthesis; review in inpatients
2. Ensure patient is on stable dietary restriction and binder if applicable.
3. Restart RAASi at half the last tolerated dose.
4. Recheck K⁺ + Cr in 2 weeks.
5. If K⁺ remains <5.5, uptitrate to full target dose.
Goal: maximum tolerated RAASi dose with K⁺ <5.5 mEq/L.
Quick Reference — Hyperkalemia + RAASi
- Default: manage K⁺ to preserve RAASi — do not reflexively stop
- K⁺ 5.5–5.9: down-titrate RAASi + add binder + dietary review
- K⁺ ≥6.0: hold RAASi + ECG + binder; restart once K⁺ <5.5
- Kalimate (CPS): 5–15 g TID in water; Ca²⁺-based — preferred in CHF/HTN; separate from other meds by 2–3 h
- Kayexalate/Resonium A (SPS): 15–60 g/day oral or enema; watch sodium load in volume-overloaded patients
- Audit list: TMP-SMX, NSAIDs, spironolactone dose, calcineurin inhibitor levels
GLP-1 Receptor Agonists — Titration & CKD Considerations
GLP-1 agonists showed kidney protection in the FLOW trial (semaglutide). The operational challenge is not starting — it is titrating through GI side effects and watching for volume-depletion-mediated AKI in CKD patients.
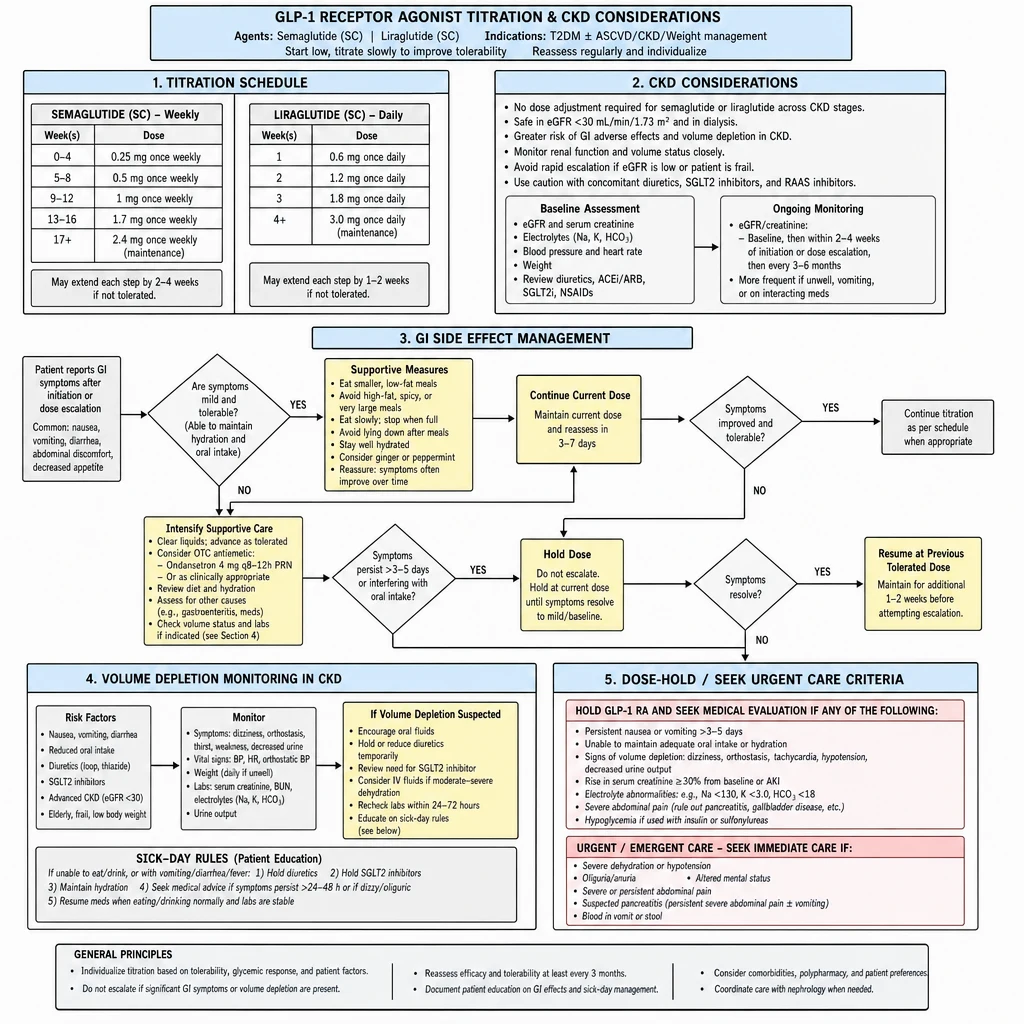 GLP-1 Agonist Titration — semaglutide/liraglutide schedule, GI management, and CKD volume monitoring.
GLP-1 Agonist Titration — semaglutide/liraglutide schedule, GI management, and CKD volume monitoring.
Titration Schedules by Agent
Semaglutide SC (Ozempic / Wegovy)
PH brands: Ozempic, Wegovy (Novo Nordisk). Refrigerate 2–8 °C; after first use, room temp <30 °C × 56 days.
Semaglutide Oral (Rybelsus)
Oral and SC semaglutide are NOT dose-interchangeable.
PH brands: Rybelsus (Novo Nordisk). Store at room temp <30 °C.
Tirzepatide (Mounjaro) — GIP + GLP-1 dual agonist
PH brands: Mounjaro (Eli Lilly). Refrigerate 2–8 °C; after first use, room temp <30 °C × 21 days.
Dulaglutide (Trulicity)
PH brands: Trulicity (Eli Lilly). Refrigerate 2–8 °C; after first use, room temp <30 °C × 14 days.
GI Side Effect Management
Avoid large, fatty, or spicy meals within 2 h of injection or oral dose.
Slower titration: stay at each dose level for 6–8 weeks instead of 4 if GI-limited.
Antiemetics: ondansetron 4–8 mg PRN or metoclopramide 5–10 mg before meals; use short-term.
Persistent vomiting >24 h → hold drug (AKI risk from dehydration; see below).
Counsel explicitly: if not tolerating fluids for >24 h, hold GLP-1 agonist and seek medical attention.
Monitor: eGFR at baseline, 4 weeks, 3 months after initiation.
Hold if: persistent epigastric pain radiating to back, amylase/lipase >3× ULN.
Discontinue permanently if confirmed pancreatitis.
Reduce concurrent insulin dose by 10–20% at initiation; reduce sulfonylurea by 50% or more to avoid hypoglycemia.
Check eGFR at 4 weeks and 3 months — GLP-1 can improve eGFR over time (FLOW data).
Hold rules — when to pause GLP-1 agonists
- Hold 24 h before elective surgery requiring general anesthesia (delayed gastric emptying → aspiration risk)
- Hold during acute illness with vomiting or inability to hydrate
- Hold if pancreatitis suspected (persistent epigastric pain)
- Hold if significant volume depletion suspected (syncope, orthostasis, Cr rising)
Quick Reference — GLP-1 Titration
- Semaglutide SC: 0.25 → 0.5 → 1.0 mg Q4 weeks (each step minimum 4 weeks)
- Bedtime injection = less daytime nausea
- No renal dose adjustment for semaglutide, tirzepatide, or dulaglutide
- Nausea + vomiting >24 h → hold drug → AKI risk in CKD from dehydration
- Surgery: hold ≥24 h pre-op (aspiration risk from delayed gastric emptying)
- Reduce co-prescribed insulin by 10–20% at initiation
Diuretic Sequencing — Loop, Thiazide & Sequential Nephron Blockade
Loop diuretic resistance is one of the most frustrating problems in managing fluid-overloaded CKD and heart failure patients. The solution is not increasing the dose indefinitely — it is sequential nephron blockade and understanding bioavailability differences between loop agents.
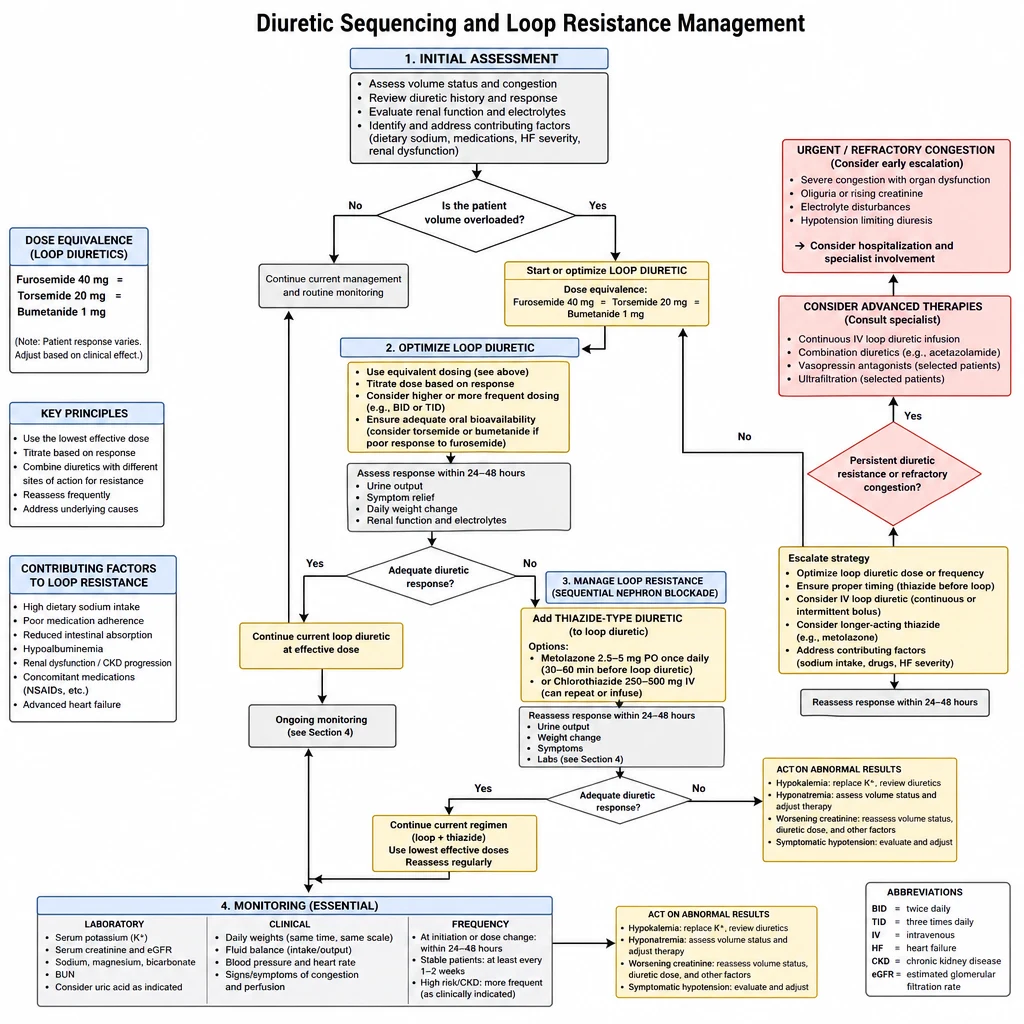 Diuretic Sequencing — loop equivalence, resistance steps, and sequential nephron blockade.
Diuretic Sequencing — loop equivalence, resistance steps, and sequential nephron blockade.
Loop Diuretic Equivalence & Bioavailability
When to switch from furosemide to torsemide
If patient has gut wall edema from fluid overload, oral furosemide absorption drops unpredictably. Torsemide's superior oral bioavailability means the switch alone — at equivalent dose — often produces markedly better diuresis. Try this before escalating dose or adding metolazone.
Furosemide: 200 mg/dose IV or PO (up to 400 mg/day) · Torsemide: 100–200 mg/day · Bumetanide: 10 mg/day
Beyond ceiling → switch agent or add thiazide; more of the same drug will not help.
Agent of choice: Metolazone 2.5–5 mg PO, given 30–60 minutes BEFORE the loop diuretic dose. PH brands: Metoz (~₱40–60/tab; limited supply — stock up). Chlorthalidone or hydrochlorothiazide 25–50 mg are alternatives when metolazone is unavailable.
Effect: can double or triple natriuresis. The combination is powerful — expect significant electrolyte losses.
Watch for:
- Hypokalemia (<3.5 mEq/L) → oral KCl supplementation; IV if <3.0 with symptoms
- Hypomagnesemia (<0.7 mEq/L) → oral Mg or IV if severe; hypomagnesemia perpetuates hypokalemia
- Hyponatremia — if Na⁺ falling acutely, reduce thiazide dose
- Pre-renal azotemia: BUN:Cr ratio >20 with rising Cr → over-diuresis → back off diuretics
Stop or slow if: JVP flat / undetectable, orthostatic hypotension, Cr rising >30%, symptomatic dizziness, goal weight achieved.
Maintenance: once dry weight achieved, switch to once-daily furosemide or torsemide at lowest effective dose.
Caution: eGFR <30 mL/min or K⁺ >4.5 → use only with careful monitoring + binder backup.
Finerenone (Kerendia): non-steroidal MRA; approved for DKD; significantly less hyperkalemia than spironolactone; safer in CKD G3–G4.
Quick Reference — Diuretic Sequencing
- Torsemide > furosemide in gut edema (better oral bioavailability)
- Double the dose before increasing frequency (hit the ceiling)
- Metolazone: 2.5–5 mg, give 30–60 min BEFORE loop diuretic
- Daily electrolytes × 3–5 days when adding thiazide
- BUN:Cr >20 + rising Cr = over-diuresis, not CKD progression
- Finerenone preferred over spironolactone in CKD + DKD
Phosphate Binder Timing Strategies
Binders only work if taken at the right time. A patient taking calcium carbonate between meals (as an antacid) gets no phosphorus binding — they just accumulate calcium. The most impactful intervention is often just fixing the timing.
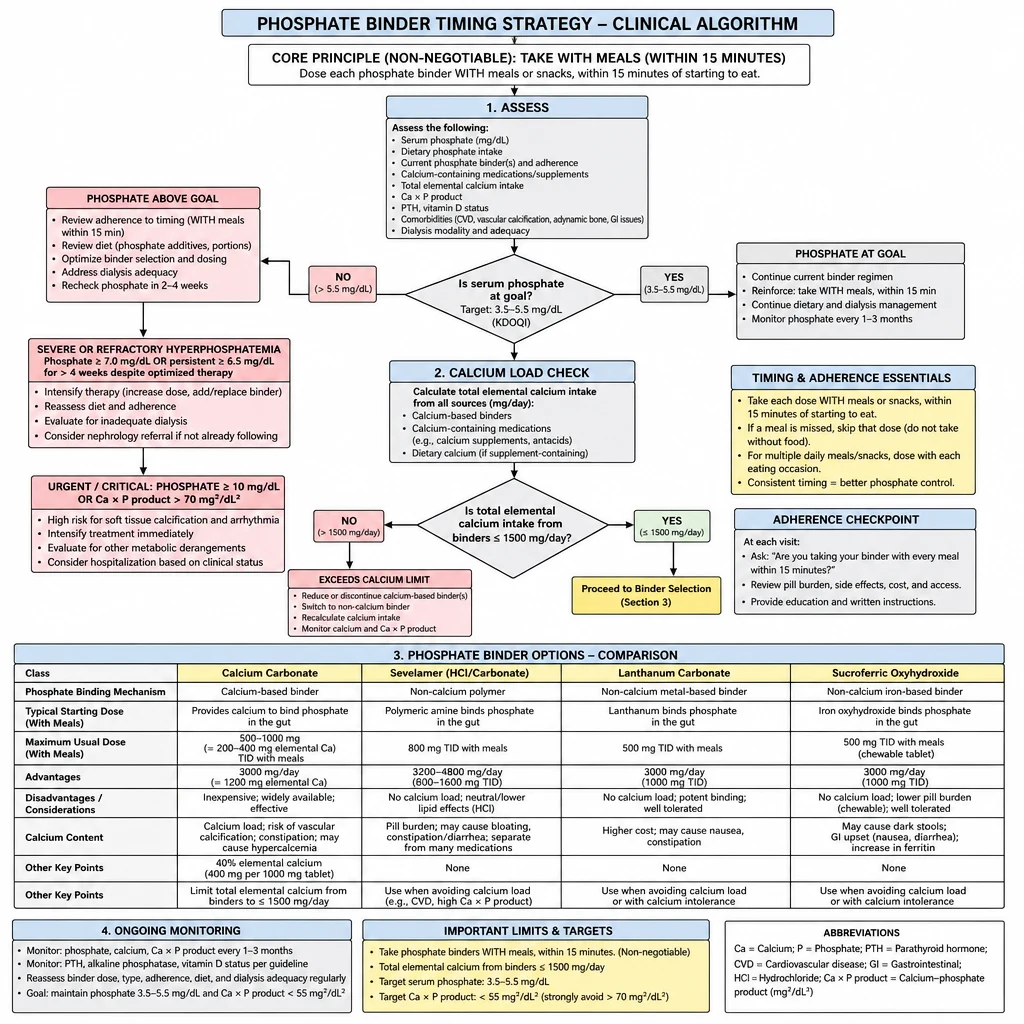 Phosphate Binder Timing — meal-based dosing rules and calcium load tracking by binder type.
Phosphate Binder Timing — meal-based dosing rules and calcium load tracking by binder type.
The non-negotiable timing rule for ALL binders
Phosphate binders must be taken with meals or immediately after (within 15 minutes). Dietary phosphorus appears in the gut lumen only when food is being digested. A binder taken 2 hours later, or between meals, binds nothing useful — it is an empty dose.
Binder-by-Binder Operational Guide
| Binder | Timing | Calcium load? | Key operational notes | Avoid if |
|---|---|---|---|---|
| Calcium carbonate (CaCO₃) PH: Caltrate, Calcimax, generic · ~₱3–8/tab |
With meals — NOT between meals | Yes — 500 mg elemental Ca per 1250 mg tab | Chew or swallow with food. Acid-suppressing drugs (PPIs) reduce efficacy. Maximum 1,500 mg/day elemental Ca from binders. | Hypercalcemia; high Ca × P product; vascular calcification on imaging |
| Calcium acetate PH: PhosLo, Acetacal, generic · ~₱10–20/tab |
With meals — NOT between meals | Yes — 169 mg elemental Ca per 667 mg tab | More phosphorus binding per mg of calcium than carbonate; 2 tabs per meal as starting dose. | Same as calcium carbonate |
| Sevelamer carbonate PH: Renvela (Sanofi) · ~₱60–90/tab |
With meals | None | 800 mg TID as starting dose; titrate to serum P. Also lowers LDL (bile acid binding). Large tablets — not suitable if dysphagia. | Bowel obstruction; dysphagia; swallowing difficulty |
| Lanthanum carbonate PH: Fosrenol (limited availability) · check pharmacy |
Chew completely with or immediately after meals | None | 500–1000 mg TID. Must chew — do not swallow whole. GI side effects common (nausea, vomiting). Strong phosphate affinity. | GI obstruction |
| Sucroferric oxyhydroxide PH: Velphoro (limited availability) · check pharmacy |
Chew with meals | None (iron-based) | 500 mg TID. Dark/black stools are expected — warn patients this is not blood. Very low iron absorption. | Iron overload; hemochromatosis |
Calcium Load Tracking
Dietary calcium: typical Filipino diet provides ~600–800 mg/day.
Binder calcium: calcium carbonate 500 mg elemental Ca per 1,250 mg tablet; 3 tabs/day = 1,500 mg.
Combined: 600 + 1,500 = 2,100 mg → over target → switch to non-calcium binder.
Levothyroxine — calcium reduces absorption → separate by ≥4 h (ideally take thyroid med morning, fasting)
Mycophenolate mofetil — sevelamer may reduce levels → take MMF ≥2 h before sevelamer
Oral iron supplements — avoid co-administration with calcium or sevelamer → separate by ≥2 h
Review the patient's snack pattern. If they eat 2+ phosphorus-significant snacks, prescribe a small binder dose (e.g., 1 tab sevelamer or half-dose CaCO₃) with those snacks.
Quick Reference — Binder Timing
- All binders: with meals or within 15 min after — never between meals
- CaCO₃ between meals = antacid, not phosphate binder
- Lanthanum: must chew completely; not swallowed whole
- Sevelamer: also lowers LDL; avoid in dysphagia/bowel obstruction
- Fluoroquinolones: separate from binders by ≥2–6 h (chelation)
- Levothyroxine: separate from calcium binders by ≥4 h
- Total calcium target: <2,000 mg/day (diet + binders combined)
ESA Adjustment Logic — Iron First, Then Dose
The single most common ESA prescribing error is escalating the dose without replenishing iron. ESAs are futile in iron deficiency — they accelerate iron utilization and deepen the deficiency. Always address iron before touching the ESA dose.
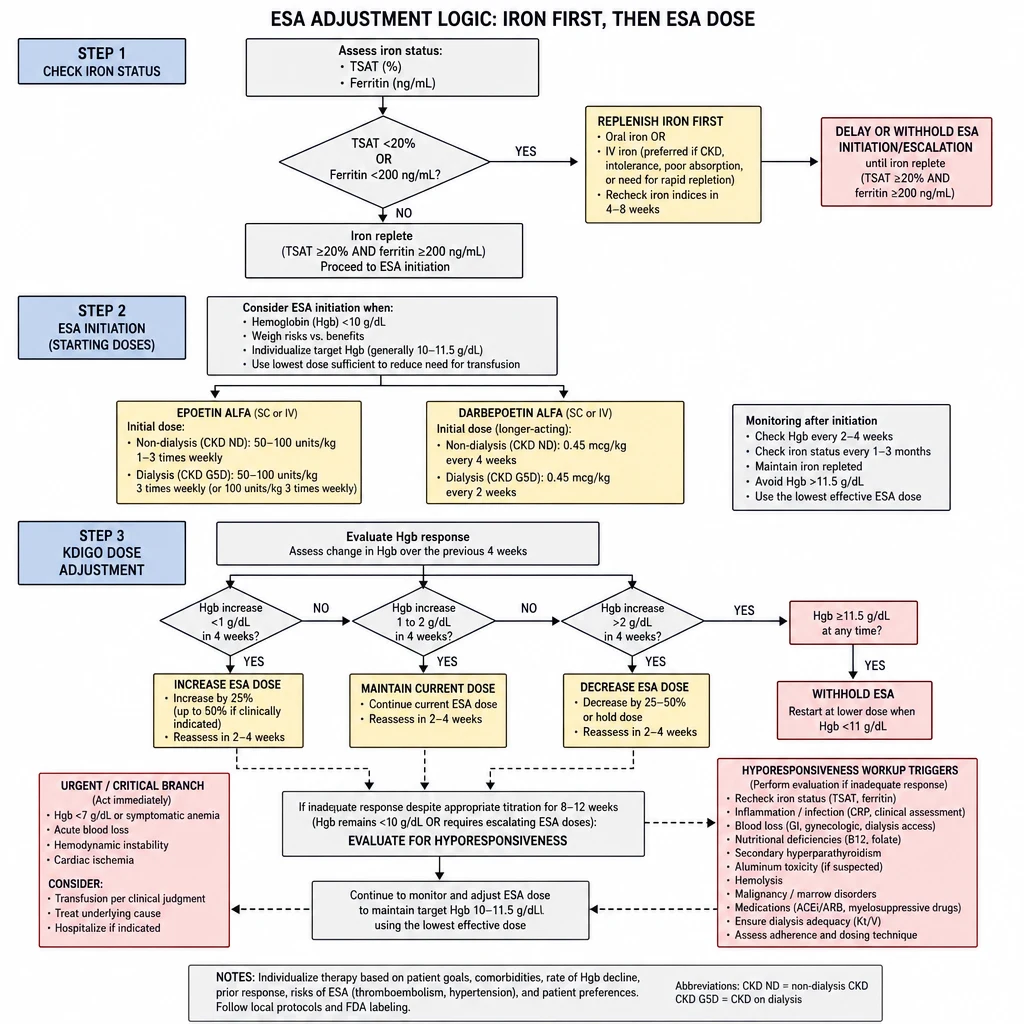 ESA Adjustment Logic — iron repletion first, KDIGO dose framework, and hyporesponsiveness workup.
ESA Adjustment Logic — iron repletion first, KDIGO dose framework, and hyporesponsiveness workup.
Iron first — always evaluate before adjusting ESA dose
If TSAT <20% or ferritin <200 ng/mL (dialysis) or <100 ng/mL (non-dialysis), replete iron before escalating ESA. Escalating ESA in iron-deficient state wastes medication and can cause functional iron deficiency with treatment failure.
Iron Repletion Before ESA
Iron Targets Before Starting/Escalating ESA
Dialysis patients: ferritin >200 ng/mL AND TSAT >20%.
Non-dialysis CKD: ferritin >100 ng/mL AND TSAT >20%.
Oral iron: ferrous sulfate 325 mg (65 mg elemental Fe) QD–TID. Take on empty stomach for best absorption. Separate from phosphate binders by ≥2 h. Common GI side effects → consider ferrous gluconate if intolerant.
IV Iron — When and Which
Preferred in: dialysis, oral intolerance, malabsorption, severe/urgent deficiency.
Iron sucrose (Venofer) 100–200 mg IV per session, over 15–30 min
Ferric carboxymaltose (Ferinject) 500–1,000 mg single IV infusion (outpatient-friendly)
Ferumoxytol 510 mg IV, may give 1,020 mg at once
Recheck iron indices (TSAT, ferritin) 4–6 weeks after completing IV iron course.
ESA Starting Doses
| Agent | Non-dialysis CKD | Hemodialysis | Peritoneal Dialysis |
|---|---|---|---|
| Epoetin alfa (Eprex, Hemapo) | 50–80 IU/kg SC 3×/week | 50–100 IU/kg SC/IV 3×/week | 50–80 IU/kg SC 3×/week |
| Darbepoetin alfa (Aranesp) | 0.45 mcg/kg SC Q4 weeks | 0.6–0.75 mcg/kg SC/IV Q2 weeks | 0.45–0.6 mcg/kg SC Q2–4 weeks |
| Mircera (C.E.R.A.) | 0.6 mcg/kg SC Q2 weeks initially | 0.6 mcg/kg SC/IV Q2 weeks initially | 0.6 mcg/kg SC Q2 weeks initially |
All: once Hgb is stable in target range (10–11.5 g/dL), Mircera may be given Q4 weeks. Reduce insulin dose if using SC epoetin (injection-site effect).
ESA Dose Adjustment Framework (KDIGO)
Rapid rise rule — reduce dose proactively
If Hgb rises >1 g/dL in 2 weeks or >2 g/dL in 4 weeks → reduce ESA by 25%, even if still in target range. Rapid rise is associated with worse outcomes (hypertension, thrombosis). A controlled, gradual rise to target is the goal.
Hyporesponsiveness Work-Up
Definition: Hgb below target despite epoetin >300 IU/kg/week (or darbepoetin >1.5 mcg/kg/week) for ≥4 weeks, with iron replete. Do NOT simply escalate dose further without work-up.
Aluminum: suspect in patients using aluminum-based antacids long-term or older water systems. Check aluminum level; treat with deferoxamine if elevated.
Further reading — ESA, iron targets, and Ganzoni calculator
For full ESA selection rationale, iron repletion protocols, hemoglobin goal evidence, and an interactive Ganzoni iron-dose calculator, see the standalone guide: Anemia Management in CKD →
Quick Reference — ESA Adjustment Logic
- Iron before ESA — always check TSAT + ferritin first
- Target Hgb:
10–11.5 g/dL— never target >13 - Hold if Hgb >13; resume at 25% lower dose when <12
- Rapid rise >1 g/dL in 2 weeks → reduce dose 25%
- Hyporesponsive: iron → CRP → Kt/V → PTH → Al → blood loss → PRCA
- PRCA (anti-ESA antibodies): stop all ESAs, test for antibodies
- IV iron: ferric carboxymaltose 500–1,000 mg single dose (convenient)
- Dialysis: recheck iron indices monthly when on ESA
Mineralocorticoid Receptor Antagonists — Finerenone vs. Spironolactone
Two very different drugs targeting the same receptor. Spironolactone is steroidal and potent — effective in HFrEF and hyperaldosteronism but carries significant hyperkalemia and hormonal side-effect risk in CKD. Finerenone is non-steroidal — purpose-built for DKD with a far better safety profile in renal patients.
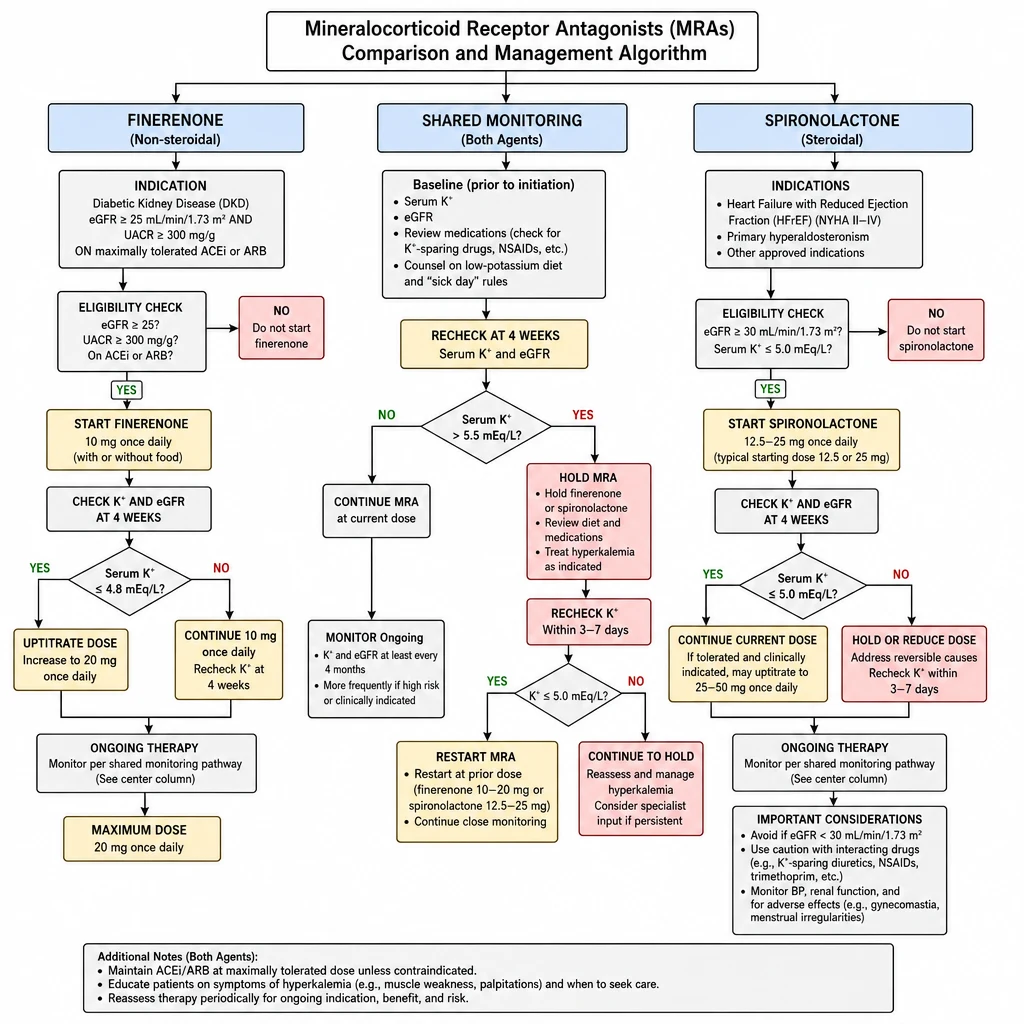 MRA Selection — Finerenone (DKD) vs Spironolactone (HFrEF), K⁺ monitoring, and dose titration.
MRA Selection — Finerenone (DKD) vs Spironolactone (HFrEF), K⁺ monitoring, and dose titration.
Finerenone (Kerendia) — non-steroidal MRA
Indication: Diabetic kidney disease with eGFR ≥25 + UACR ≥300 mg/g, on maximally tolerated ACEi or ARB.
Start dose: 10 mg OD with food. Recheck K⁺ and eGFR at 4 weeks.
Uptitrate to: 20 mg OD if K⁺ ≤4.8 mEq/L at 4-week check.
Hold if: K⁺ >5.0 mEq/L before starting; hold and recheck if K⁺ rises >5.5 on therapy.
CYP3A4 substrate: avoid grapefruit juice; avoid strong 3A4 inhibitors (azole antifungals, clarithromycin). Dose-halve with moderate 3A4 inhibitors if unavoidable.
Advantages over spironolactone in CKD: ~40% less hyperkalemia; no gynecomastia or menstrual effects; no progesterone/androgen receptor cross-reactivity.
PH brands: Firialta (Bayer) · ~₱350–450/tab · No refrigeration.
Spironolactone (Aldactone) — steroidal MRA
Indications: HFrEF with EF ≤35% (RALES/EMPHASIS-HF mortality benefit); resistant hypertension; primary hyperaldosteronism; ascites in cirrhosis; K⁺-sparing diuresis.
Start dose: 12.5–25 mg OD in CKD. Titrate to 50 mg OD (HFrEF) if K⁺ tolerated.
Use with caution if: eGFR <30 (significant K⁺ accumulation risk); K⁺ >4.5 at baseline; on other K⁺-retaining agents.
Avoid if: eGFR <30 without close monitoring + binder backup; K⁺ ≥5.0 at baseline.
Side effects: Gynecomastia and breast tenderness (men); menstrual irregularity (women) — dose-dependent; switch to eplerenone if intolerable (less hormonal SE, less evidence in HFrEF).
PH brands: Aldactone (Pfizer), generic · ~₱15–30/tab · No refrigeration.
Which Agent — Decision Guide
| K⁺ on therapy | Action |
|---|---|
| ≤5.0 mEq/L | Continue; uptitrate finerenone to 20 mg if still at 10 mg |
| 5.0–5.5 mEq/L | Reduce dose by 50%; dietary K⁺ review; add binder if needed |
| >5.5 mEq/L | Hold; ECG; manage hyperkalemia; restart at lower dose once K⁺ <5.0 |
Quick Reference — MRA Selection
- Finerenone: DKD + eGFR ≥25 + UACR ≥300 + on max ACEi/ARB
- Start finerenone only if K⁺ ≤4.8; uptitrate 10→20 mg at 4 weeks if K⁺ ≤4.8
- CYP3A4 substrate: avoid azoles, clarithromycin, grapefruit
- Spironolactone: HFrEF EF ≤35%, resistant HTN, primary hyperaldosteronism
- Spironolactone: caution eGFR <30; avoid K⁺ >5.0 baseline
- Never combine finerenone + spironolactone
Anticoagulation in CKD — DOAC Selection & Dose Adjustment
CKD patients have both increased bleeding and increased clotting risk — the management paradox. DOAC selection hinges on the degree of renal clearance of each agent. Dabigatran is the most dangerous in CKD; apixaban has the least renal exposure and is generally preferred.
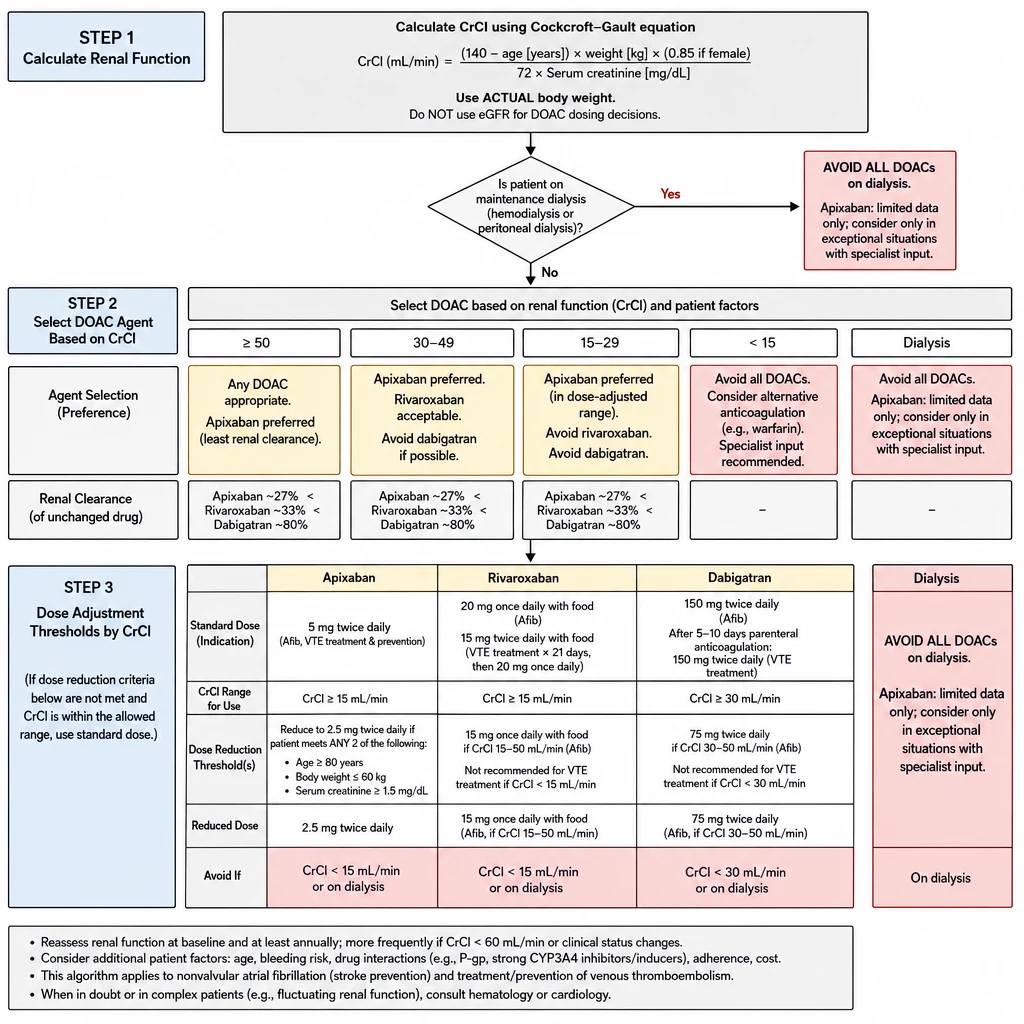 DOAC Selection in CKD — CrCl-based agent choice, apixaban preference, and dose reduction thresholds.
DOAC Selection in CKD — CrCl-based agent choice, apixaban preference, and dose reduction thresholds.
Use CrCl (Cockcroft-Gault), not eGFR, for DOAC dosing
All DOAC product inserts use CrCl by Cockcroft-Gault for dose adjustment — not CKD-EPI eGFR. In elderly or low-muscle-mass patients, CrCl may be significantly lower than eGFR. Always calculate CrCl before selecting DOAC dose.
DOAC Comparison — Renal Clearance & CKD Dosing
| Agent | Renal clearance | Standard dose (AF) | CKD dose adjustment | PH brands |
|---|---|---|---|---|
| Apixaban Preferred in CKD |
~27% | 5 mg BID | Reduce to 2.5 mg BID if ≥2 of: age ≥80, weight ≤60 kg, SCr ≥1.5 mg/dL. Emerging data support use in dialysis. | Eliquis (BMS/Pfizer) ~₱80–110/tab |
| Rivaroxaban | ~35% | 20 mg OD with evening meal | Reduce to 15 mg OD if CrCl 15–50 mL/min. Avoid if CrCl <15. | Xarelto (Bayer/J&J) ~₱120–160/tab |
| Edoxaban | ~50% | 60 mg OD | Reduce to 30 mg OD if CrCl 15–50 mL/min, weight ≤60 kg, or P-gp inhibitor co-use. Avoid CrCl <15. | Lixiana (Daiichi) ~₱130–170/tab |
| Dabigatran Avoid in CKD G4–5 |
80–85% | 150 mg BID | Use 110 mg BID if CrCl 30–50 + high bleed risk. Avoid if CrCl <30. Not for dialysis. Highest accumulation risk of all DOACs. | Pradaxa (Boehringer) ~₱90–130/tab |
Warfarin in CKD — When & How
When warfarin remains the choice
Mechanical heart valves: DOACs are contraindicated — warfarin only. Target INR 2.5–3.5 (mitral) or 2.0–3.0 (aortic bileaflet).
Dialysis + AF (where apixaban data insufficient): warfarin remains standard in some centres, though calciphylaxis and vascular calcification risk are concerns.
Antiphospholipid syndrome (triple positive): warfarin is superior to DOACs; target INR 2.5–3.5 in high-risk thrombotic APS.
PH brands: Coumadin (BMS), generic warfarin · ~₱5–15/tab.
CKD-specific warfarin challenges
Sensitivity: CKD patients require lower warfarin doses — reduced vitamin K intake, impaired hepatic metabolism, altered protein binding.
Calciphylaxis risk: warfarin inhibits matrix Gla protein (vitamin K-dependent) — promotes vascular and soft tissue calcification. In CKD G4–5 and dialysis patients with CaxP above target, consider DOAC or warfarin minimization.
TTR target: time in therapeutic range (INR 2–3) should be >70%. Poor TTR increases both stroke and bleeding risk. If TTR <65% consistently → switch to apixaban if eligible.
Drug interactions: antibiotics (especially metronidazole, fluoroquinolones), antifungals, statins all potentiate warfarin. Recheck INR 5–7 days after any antibiotic course.
Heparin & LMWH in CKD
Apixaban 2.5 mg BID — emerging observational data; preferred by many nephrologists over warfarin in dialysis AF.
Warfarin — traditional choice; concern for calciphylaxis and coronary calcification acceleration.
Dabigatran / Rivaroxaban / Edoxaban — avoid in dialysis (accumulation, no reversal data).
Decision should factor CHA₂DS₂-VASc score, HAS-BLED score, and patient-specific calcification burden.
Quick Reference — Anticoagulation in CKD
- Use Cockcroft-Gault CrCl — not eGFR — for DOAC dosing
- Apixaban: lowest renal clearance (27%); preferred DOAC in CKD
- Dabigatran: 80–85% renal — avoid if CrCl <30
- Warfarin: still required for mechanical valves and triple-positive APS
- UFH: not renally cleared; safe in any CrCl, AKI, or dialysis
- Enoxaparin in CrCl <30: 1 mg/kg q24h + anti-Xa monitoring
- Dialysis + AF: apixaban 2.5 mg BID emerging preference over warfarin
- Warfarin: watch for calciphylaxis risk in CKD G4–5 + dialysis
Anti-Hyperglycemic Drugs — CKD to Dialysis Transition
As eGFR falls from G3b through G5 into dialysis, almost every non-insulin glucose-lowering drug requires dose reduction, substitution, or outright cessation. Insulin becomes the cornerstone — but its own dosing must be reduced by up to 50% before dialysis starts, then recalibrated again based on modality (HD vs. PD behave oppositely).
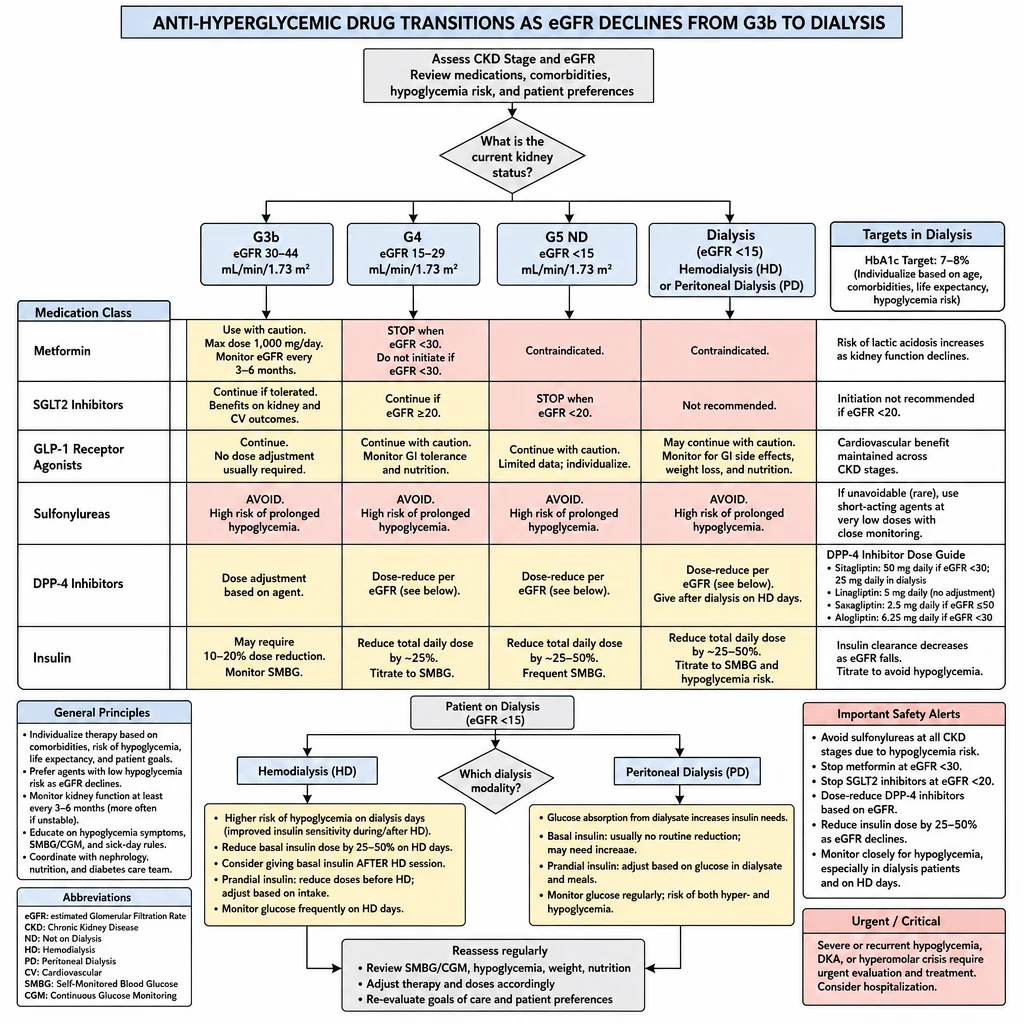 Anti-Hyperglycemic Transitions — stop/adjust/continue matrix from G3b through dialysis.
Anti-Hyperglycemic Transitions — stop/adjust/continue matrix from G3b through dialysis.
Two principles that override all others
1. Hypoglycemia is more dangerous in CKD/dialysis — reduced renal gluconeogenesis, unpredictable oral intake, drug accumulation. Target HbA1c 7–8% (not <7%) in dialysis; avoid aggressive lowering.
2. HbA1c is unreliable in dialysis — shortened red cell lifespan falsely lowers it. Use fructosamine or glucose logs alongside HbA1c in G5D patients.
Transition Matrix — Stop / Adjust / Continue / Shift
| Drug class | G3b eGFR 30–44 |
G4 eGFR 15–29 |
G5 pre-dialysis eGFR <15 |
G5D — HD | G5D — PD |
|---|---|---|---|---|---|
| Metformin | Reduce to ≤1 g/day; monitor eGFR q3mo | STOP (eGFR <30) | STOP | STOP | STOP |
| Glibenclamide | STOP — active metabolites accumulate | STOP | STOP | STOP | STOP |
| Gliclazide MR | Continue; max 60 mg/day; monitor closely | Reduce to 30 mg; use with caution | STOP | STOP | STOP |
| Glimepiride | Reduce dose; active metabolite renally cleared | STOP | STOP | STOP | STOP |
| Linagliptin | Continue 5 mg OD — no adjustment | Continue 5 mg OD | Continue 5 mg OD | Continue 5 mg OD | Continue 5 mg OD |
| Sitagliptin | Reduce to 50 mg OD (eGFR <45) | Reduce to 25 mg OD | 25 mg OD | 25 mg OD — give after HD session | 25 mg OD |
| Vildagliptin | 50 mg OD (not BID) if eGFR <50 | 50 mg OD | 50 mg OD | 50 mg OD | 50 mg OD |
| SGLT2i | Continue if eGFR ≥20 | Glycemic benefit lost; continue only for CV/HF benefit if eGFR ≥20 | STOP (eGFR <20) | STOP | STOP |
| GLP-1 agonists | Continue — no renal dose adjustment | Continue | Continue; monitor volume status | Continue — no adjustment | Continue — no adjustment |
| Pioglitazone | Continue if no edema/CHF | Caution — fluid retention; avoid if volume-overloaded | Avoid — incompatible with fluid targets | Avoid | Avoid |
| Repaglinide | Continue — biliary excretion; no renal clearance | Continue; start low (0.5 mg) | Continue with monitoring | Continue; skip dose if meal skipped | Continue; skip dose if meal skipped |
| Insulin | Reduce TDD by 15–25% | Reduce TDD by 25–40% | Reduce TDD by 40–50% | Further adjust — see HD protocol below | Usually increase — see PD protocol below |
Drug-Class Notes
PH brands: Glucophage (Merck), generic · ~₱3–8/tab.
Glimepiride (Amaryl) — active M1 metabolite; renally excreted. Reduce dose at G3b; stop at G4.
Gliclazide MR (Diamicron MR) — inactive metabolites; safest SU in CKD. Max 60 mg at G3b; reduce to 30 mg at G4; stop at G5. Still carries hypoglycemia risk — skip dose if meal is missed.
On dialysis: all sulfonylureas avoided — irregular meal patterns + uremic anorexia = unpredictable hypoglycemia risk.
Sitagliptin (Januvia) — 87% renally excreted; dose-reduce to 50 mg at eGFR <45, 25 mg at eGFR <30/dialysis. On HD: give after each session (dialysed partially).
Vildagliptin (Galvus) — 85% renally excreted; reduce to 50 mg OD (skip afternoon dose) when eGFR <50.
PH brands: Trajenta (Boehringer) ~₱60–80/tab · Januvia (MSD) ~₱70–90/tab · Galvus (Novartis) ~₱55–75/tab.
Key rule: take only with meals; skip the dose if the patient skips the meal — critical in HD patients with variable appetite on dialysis days.
Start: 0.5 mg before each main meal. Titrate to 1–2 mg TID if needed.
PH brands: NovoNorm (Novo Nordisk), Prandin · ~₱25–40/tab.
Insulin — The Cornerstone at G5 and Beyond
Avoid NPH in G4–G5 — use analogue basal insulin instead
NPH (Humulin N, Insulatard) has erratic absorption, a pronounced peak, and accumulates in advanced CKD → unpredictable prolonged hypoglycemia. Switch to glargine (Lantus / Toujeo), detemir (Levemir), or degludec (Tresiba) before the patient reaches G4.
Insulin dose reduction as eGFR declines
Why doses fall: (1) Kidney clears ~25–40% of circulating insulin. (2) Renal gluconeogenesis declines → less counterregulation. (3) Uremic toxins reduce insulin resistance modestly in late CKD.
G3b: Reduce total daily dose (TDD) by ~15–25%.
G4: Reduce TDD by 25–40%.
G5 pre-dialysis: Reduce TDD by 40–50%.
Reduce basal and prandial proportionally. Recheck glucose logs every 1–2 weeks during each transition. Snack before bed if fasting glucose trending low.
Insulin agent selection in CKD
Glargine U-100 (Lantus) OD — peakless, predictable; ~₱700–900/vial
Glargine U-300 (Toujeo) OD — longer, flatter profile; fewer nocturnal hypos; ~₱1,200–1,500/pen
Degludec (Tresiba) OD — ultra-long (42 h); most stable in dialysis; ~₱1,400–1,800/pen
Detemir (Levemir) OD–BID — flexible; ~₱900–1,100/pen
Aspart (NovoRapid) · Lispro (Humalog) — prandial analogs; safe at any eGFR
NPH (Humulin N) — avoid G4–5; erratic peak accumulates
Insulin on Dialysis — HD vs. PD Behave Oppositely
Hemodialysis (HD) — insulin requirements usually FALL
Mechanism: Standard HD dialysate contains glucose ~100 mg/dL. During a 4-hour session, glucose diffuses into the patient → BG rises during session but the overall stimulation of gluconeogenesis is less.
Hypoglycemia risk during HD: If patient is fasting before the session (or vomiting from uremia), dialysis removes glucose and provides no nutritional input. BG can fall dangerously mid-session.
Practical rules:
• Monitor BG at start, at 2 hours, and at session end.
• Reduce or omit prandial insulin for the meal immediately before a morning HD session.
• Do NOT give long-acting insulin dose right before HD without a meal planned.
• Post-HD rebound hyperglycemia is common (catecholamine effect + fluid shifts) — do not over-correct; it self-resolves.
• Adjust basal dose down 20–30% on HD days if recurrent intra-dialytic hypoglycemia.
Peritoneal Dialysis (PD) — insulin requirements usually RISE
Mechanism: PD dialysate contains dextrose (1.5%, 2.5%, or 4.25%). A 2-L bag of 4.25% dextrose delivers ~68 g glucose — significant caloric and glycemic load absorbed continuously.
Hyperglycemia is the major challenge — not hypoglycemia. CAPD patients often need MORE insulin than pre-dialysis, especially with high-dextrose bags.
Two routes for insulin in PD:
Subcutaneous (SC) — standard, as usual; titrate up with PD start.
Intraperitoneal (IP) — add regular (short-acting) insulin directly to PD bag before instillation. Only regular insulin (not analogs) for IP route. IP dose is typically 25–50% higher than equivalent SC dose (peritoneal absorption is slower). Use aseptic technique — infection risk.
Icodextrin bags: icodextrin (Extraneal, used for long dwell) is a glucose polymer — falsely elevates BG on some glucometers (electrochemical strips). Use glucose oxidase-based meters (e.g., FreeStyle series) — not GDH-PQQ-based strips.
Suggested Transition Protocol
Quick Reference — Anti-Hyperglycemic Transition
- Stop metformin at eGFR <30 (lactic acidosis)
- Stop glibenclamide at G3b; stop all SUs by G5
- Linagliptin: 5 mg OD at any eGFR — no adjustment ever
- SGLT2i: stop when eGFR <20; no mechanism on dialysis
- GLP-1 agonists: continue through dialysis — no renal dose adjustment
- Repaglinide: safe at any stage; skip dose if meal skipped
- Insulin TDD: −25% at G3b → −40% at G4 → −50% at G5
- Avoid NPH in G4–5 — switch to glargine, detemir, or degludec
- HD: reduce/omit prandial insulin before morning session; monitor mid-session BG
- PD: glucose load from dialysate → insulin needs RISE; consider IP insulin
- IP insulin: regular insulin only (not analogs); dose 25–50% higher than SC
- HbA1c unreliable in dialysis — use fructosamine or 14-day BG log
Sleep Disturbance in CKD and ESKD
Sleep disorders affect 50–80% of dialysis patients and are among the strongest independent predictors of quality of life, cardiovascular mortality, and dialysis withdrawal. They are systematically under-asked and under-treated. The four disorders that dominate are restless legs syndrome, sleep apnea, insomnia, and excessive daytime sleepiness — each with a different treatment pathway.
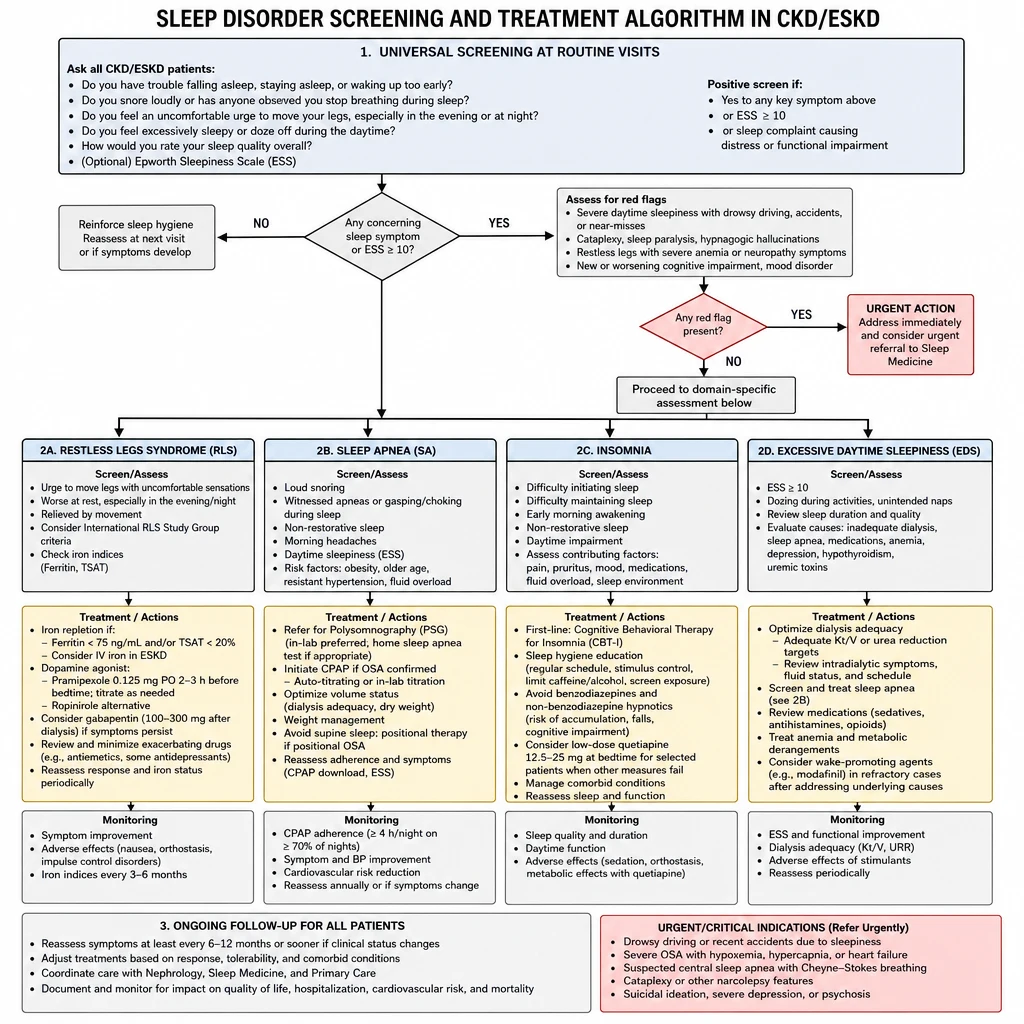 Sleep Disorders in CKD/ESKD — screening questions and four-pathway treatment algorithm.
Sleep Disorders in CKD/ESKD — screening questions and four-pathway treatment algorithm.
Ask first — most patients never volunteer sleep complaints
Screen every dialysis patient at each clinic visit with three questions: "Do you have an urge to move your legs at night?" · "Has anyone told you that you stop breathing during sleep?" · "How many hours do you sleep — do you feel rested when you wake?" A positive screen on any directs the workup below.
Sleep Disorder Types in CKD/ESKD — Overview
| Disorder | Prevalence in dialysis | Key feature | First step |
|---|---|---|---|
| Restless Legs Syndrome (RLS) | 20–40% | Irresistible urge to move legs at night; worse at rest; relieved by movement | Check iron, optimize dialysis adequacy |
| Obstructive Sleep Apnea (OSA) | 30–50% | Snoring, witnessed apneas, morning headache, EDS | Polysomnography; CPAP |
| Central Sleep Apnea (CSA) | 10–30% | Cheyne-Stokes pattern; common in HFrEF + ESKD | Optimize HF management; consider nocturnal HD |
| Insomnia | 50–70% | Difficulty initiating or maintaining sleep; early waking | Sleep hygiene + CBT-I before pharmacotherapy |
| Excessive Daytime Sleepiness | 40–60% | Epworth scale ≥10; often secondary to above disorders | Treat underlying primary disorder |
Restless Legs Syndrome — CKD-Specific Management
Dialysis adequacy: Kt/V <1.2 worsens uremic RLS. Optimize dialysis dose. Nocturnal HD (6 nights/week) dramatically reduces RLS burden — consider referral if standard HD is failing.
Drug audit: remove aggravating drugs — antihistamines (diphenhydramine, hydroxyzine), metoclopramide, antipsychotics, antidepressants (especially SSRIs, TCAs), caffeine.
CKD dose adjustment: pramipexole is 90% renally excreted. In CrCl <20: 0.125 mg OD (do not exceed). In dialysis: 0.125 mg every other day or 3×/week — give on non-dialysis evenings if possible.
Ropinirole (Requip) — start 0.25 mg at bedtime; titrate to 0.5–2 mg over 2–4 weeks. Primarily hepatic metabolism — safer than pramipexole in dialysis; no strict renal dose adjustment required.
Augmentation: long-term dopamine agonist use can cause paradoxical worsening (RLS earlier in day, spreading to arms). If augmentation develops, switch to an alpha-2-delta ligand (Step 3).
PH brands: Sifrol (pramipexole, Boehringer) · ~₱30–60/tab.
Pregabalin — same principle as gabapentin. Post-HD dosing: 25–75 mg after each session. Also avoid in PD for same reason. Both have sedative, dizziness, and fall risk — start low.
PH brands: Neurontin / generic gabapentin · ~₱15–35/cap · Generic pregabalin, Lyrica (Pfizer) · ~₱30–60/cap.
Manage by: gradually tapering dopamine agonist (do not stop abruptly — rebound) while cross-titrating to gabapentin/pregabalin post-HD. If refractory, low-dose opioids (tramadol, codeine — use with caution in CKD) have evidence.
Sleep Apnea in ESKD
Obstructive Sleep Apnea (OSA)
Diagnosis: polysomnography or home sleep apnea test (HSAT). Refer to pulmonology/sleep medicine for suspected moderate–severe OSA (AHI ≥15 or symptomatic AHI ≥5).
Treatment: CPAP is the primary therapy — reduces AHI, improves daytime sleepiness, may reduce cardiovascular events. Auto-titrating CPAP (APAP) is acceptable for uncomplicated OSA.
Volume overload contributes: excess fluid redistributes to the neck during recumbency → upper airway narrowing. Aggressive fluid removal (optimized UF targets) can reduce OSA severity in HD patients. Elevate head of bed ≥30°.
Central Sleep Apnea / Cheyne-Stokes Respiration
Context: CSA with Cheyne-Stokes breathing is most common in the setting of HFrEF + ESKD — the combination of impaired cardiac output and uremia creates loop gain instability in respiratory control.
Treat the HF: optimizing cardiac function with guideline-directed therapy (ACEi/ARB, beta-blockers, SGLT2i, MRA) reduces CSA severity.
Nocturnal HD: multiple studies show nocturnal HD (5–6 nights/week, 6–8 h/session) significantly reduces both OSA and CSA burden — likely via improved fluid balance and uremic solute clearance. Consider nephrology referral for nocturnal HD in patients with refractory sleep apnea.
ASV caution: adaptive servo-ventilation is contraindicated in HFrEF with EF ≤45% (SERVE-HF trial: increased mortality).
Insomnia — Non-Pharmacologic First, Then Pharmacotherapy
Sleep hygiene for dialysis patients specifically:
- Schedule dialysis to avoid sessions ending late evening — post-HD arousal state impairs sleep onset
- Fixed wake time 7 days/week — anchors circadian rhythm
- Avoid napping >20 min after 2 pm
- Bright light exposure in the morning; dim lights 2 h before bed
- Address nocturia — if post-HD fluid shifts cause nocturia, adjust target weight and UF strategy
| Agent | Dose | CKD safety | Notes |
|---|---|---|---|
| Melatonin | 3–5 mg at bedtime | Safe at any eGFR; no dose adjustment | OTC; minimal side effects; best for circadian misalignment; start here. PH: available OTC ~₱20–40/tab. |
| Mirtazapine | 7.5–15 mg at bedtime | Use with caution in G4–5; reduce dose; sedation is the target effect | Useful when insomnia co-exists with depression or anorexia (stimulates appetite). Avoid if drowsy during dialysis sessions. PH: generic, Remeron (Organon) ~₱30–50/tab. |
| Low-dose doxepin | 3–6 mg at bedtime | Caution in CKD — metabolite accumulation; reduce dose; avoid G5 | Approved specifically for sleep maintenance insomnia. Anticholinergic effects; falls risk. Use only if melatonin + mirtazapine fail. |
| Clonazepam | 0.25–0.5 mg at bedtime | Accumulates in CKD; start at 0.25 mg; short-term use only (≤4 weeks) | Useful for RLS + insomnia combination. Risk: respiratory depression, dependence, falls in elderly. Avoid in OSA. PH: generic, Rivotril (Roche) ~₱15–30/tab. |
| Zolpidem / Zopiclone | — | Avoid in CKD G4–5 and elderly dialysis patients | Accumulation → prolonged sedation, falls, delirium. If used, halve dose and limit to 2–3 doses/week. |
| Diphenhydramine (antihistamine) | — | Avoid — worsens RLS; anticholinergic delirium risk | Commonly in OTC sleep aids (e.g., Benadryl). Explicitly contraindicates in any patient with RLS or CKD G4–5. |
Dialysis-Specific Interventions That Improve Sleep Across All Disorder Types
Optimize dialysis adequacy
Kt/V <1.2 worsens uremic RLS, insomnia, and EDS. A single rise in Kt/V from 1.0 to 1.4 produces measurable sleep improvement. Check Kt/V every 1–3 months. Uremic pruritus (a major sleep disruptor) also improves with better adequacy.
Correct anemia and iron
Target Hgb 10–11.5 g/dL. Iron deficiency independently worsens RLS regardless of anemia status. IV iron repletion (TSAT >20%, ferritin >200 in dialysis) should precede any RLS pharmacotherapy. Fatigue from anemia overlaps heavily with insomnia and EDS — treating one often improves the other.
Nocturnal HD for refractory cases
Patients on nocturnal HD (5–6×/week, 6–8 h overnight) consistently report dramatic sleep quality improvement — likely due to continuous uremic solute clearance, better fluid control, and less post-HD sympathetic arousal. If a patient's sleep disorders are driving poor quality of life despite all other interventions, nocturnal HD referral is warranted.
Quick Reference — Sleep Disturbance in CKD/ESKD
- Screen every dialysis patient: RLS · apnea · insomnia
- RLS Step 1: IV iron (if TSAT <20%) + optimize Kt/V
- RLS Step 2: pramipexole 0.125 mg at bedtime (0.125 mg every other day in dialysis)
- RLS Step 3: gabapentin 100–300 mg post-HD only (never in PD)
- OSA: CPAP; address fluid overload; UF optimization
- CSA + HFrEF: treat HF first; nocturnal HD if refractory; ASV contraindicated if EF ≤45%
- Insomnia: CBT-I first; melatonin 3–5 mg (safest drug); mirtazapine 7.5 mg if co-morbid depression
- Avoid: diphenhydramine (worsens RLS), zolpidem in G4–5 (accumulates), benzodiazepines long-term
- Gabapentin/pregabalin: post-HD dosing only; never give pre-HD or in PD
- Augmentation on dopamine agonists: cross-titrate to gabapentin post-HD
- HbA1c is unreliable when managing glycemia + sleep in dialysis — use fructosamine
- Nocturnal HD improves RLS, OSA, CSA, and insomnia — most effective non-drug intervention
Antibiotic Stewardship in CKD and ESKD
CKD patients are caught between two forces: they are more susceptible to infection (uremic immune dysfunction, dialysis access, frequent healthcare contact) and more vulnerable to antibiotic toxicity (drug accumulation, nephrotoxin-on-injured-kidney, hyperkalemia from TMP-SMX). Stewardship in this population means choosing the right drug, the right dose, and the right duration — and knowing which common prescribing habits are quietly dangerous.
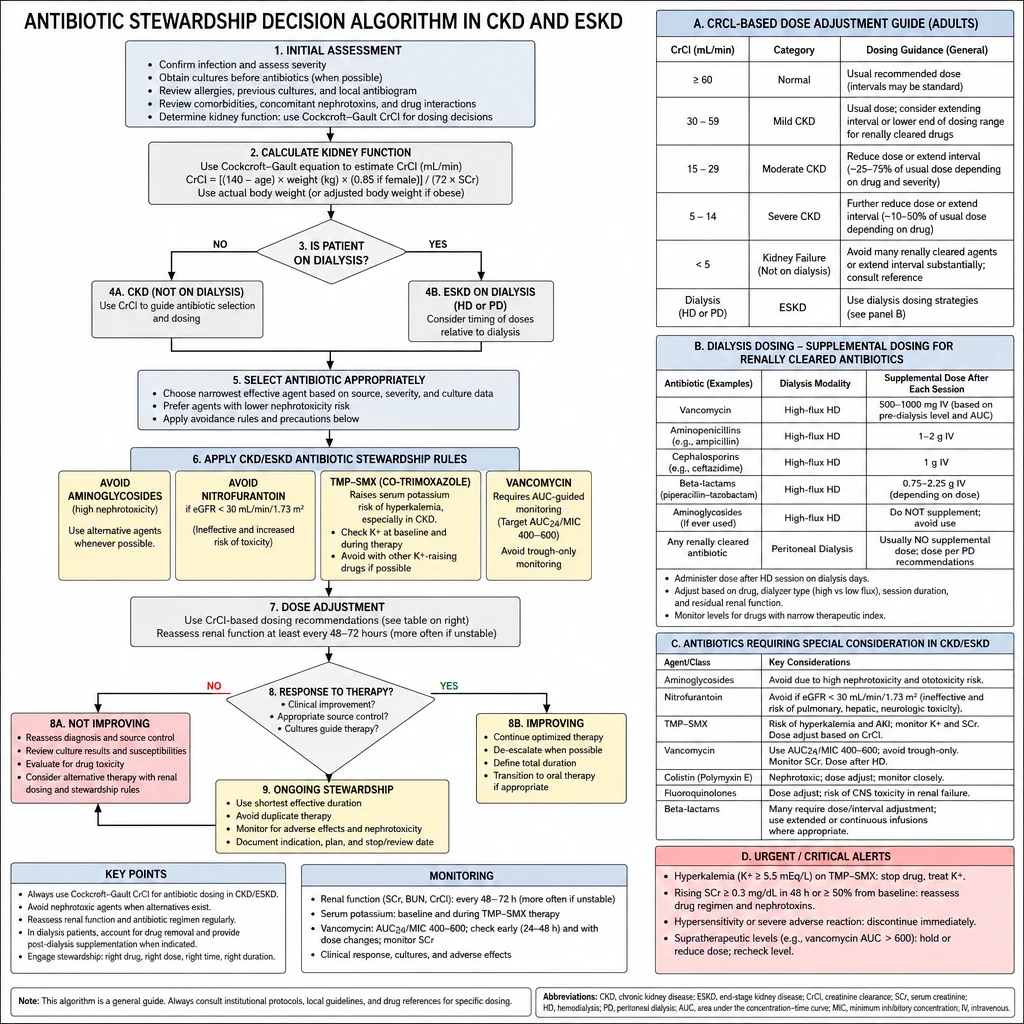 Antibiotic Stewardship in CKD — CrCl-based dosing, nephrotoxin avoidance, and dialysis supplement rules.
Antibiotic Stewardship in CKD — CrCl-based dosing, nephrotoxin avoidance, and dialysis supplement rules.
Use Cockcroft-Gault CrCl — not eGFR — for antibiotic dose adjustment
All antibiotic dosing references use CrCl by Cockcroft-Gault. In elderly, low-muscle-mass, or malnourished patients the eGFR (CKD-EPI) overestimates renal drug clearance. A 70-year-old, 50 kg woman with creatinine 1.2 mg/dL may have CrCl ~28 mL/min despite an eGFR suggesting G3a. Always calculate CrCl before selecting dose.
Core Stewardship Principles in CKD
Loading dose vs. maintenance dose
Loading dose: generally unchanged — determined by volume of distribution (Vd), not clearance. Skipping or reducing the loading dose leads to delayed therapeutic levels. Give full loading dose even in dialysis.
Maintenance dose: this is where renal adjustment applies — either reduce the dose, extend the interval, or both. The choice depends on the drug's toxicity profile: narrow therapeutic index drugs (vancomycin, aminoglycosides) require TDM; broad-index drugs (amoxicillin) tolerate interval extension alone.
Culture first; de-escalate early
Dialysis patients carry resistant organisms (MRSA, ESBL-producing Enterobacteriaceae, VRE) at higher prevalence due to repeated healthcare exposure. Empiric coverage must reflect this. But empiric broad-spectrum therapy should be de-escalated within 48–72 h once culture results are available — continuing meropenem for a susceptible E. coli just because the patient "looks sick" is stewardship failure.
Duration: use the shortest evidence-based course. 3 days for uncomplicated lower UTI (women); 5–7 days for pyelonephritis; 14 days for bacteraemia (not 21 unless source control problem); 6 weeks for osteomyelitis.
Antibiotic Dose Adjustment Reference — CKD and Dialysis
| Antibiotic | Normal dose | CrCl 30–60 | CrCl 10–29 | HD supplement | PH brands |
|---|---|---|---|---|---|
| Beta-lactams | |||||
| Amoxicillin-clavulanate | 875/125 mg q8h | 875/125 mg q12h | 500/125 mg q12h | One extra dose after HD | Augmentin (GSK), generic |
| Cefazolin IV | 1–2 g q8h | 1 g q12h | 1 g q24h | 1 g after each HD session — preferred agent for HD access infections (MSSA) | Generic, widely available |
| Ceftriaxone IV/IM | 1–2 g q24h | No adjustment | No adjustment | No supplement needed (biliary clearance) | Rocephin (Roche), generic |
| Cefepime IV Neurotox risk | 1–2 g q8–12h | 1 g q12h | 500 mg q24h | 1 g after each HD session | Maxipime, generic |
| Pip-tazo IV | 4.5 g q6–8h | 3.375 g q8h | 2.25 g q8h | 0.75 g after each HD session | Tazocin (Pfizer), generic |
| Meropenem IV | 1–2 g q8h | 1 g q12h | 500 mg q12h; 500 mg q24h if CrCl <10 | 500 mg after HD if dose given within 6 h of session end | Meronem (AstraZeneca), generic |
| Ertapenem IV | 1 g q24h | No adjustment | 500 mg q24h (CrCl <30) | 150 mg supplement if HD within 6 h of dose | Invanz (MSD) |
| Fluoroquinolones | |||||
| Ciprofloxacin oral | 500 mg q12h | 250–500 mg q12h | 250 mg q12h | Give after HD; some cleared by dialysis | Ciprobay (Bayer), generic |
| Levofloxacin oral/IV | 500 mg q24h | 250 mg q24h | 250 mg q48h (or 500 mg loading → 250 mg q48h) | No supplement (not significantly dialysed) | Cravit (Sanofi), generic |
| Moxifloxacin oral | 400 mg q24h | No adjustment | No adjustment | No supplement (biliary — useful when renal adjustment impractical) | Avelox (Bayer) |
| Glycopeptides | |||||
| Vancomycin IV TDM required | 15–20 mg/kg q8–12h | 15–20 mg/kg q12–24h; use TDM | 15–20 mg/kg loading; redose by trough or AUC | Redose 500 mg–1 g when trough <15 (typically q3–7 days); high-flux membranes remove more | Generic, Vancocin |
| Teicoplanin IV/IM | 6 mg/kg q12h ×3 then q24h | q48h after day 4 | q72h after day 4 | Not significantly dialysed; no supplement | Targocid (Sanofi) |
| Other key agents | |||||
| TMP-SMX oral K⁺ risk | 1 DS tab q12h | 1 DS tab q12h; check K⁺ | Use with caution; 1 DS q24h; check K⁺ closely; avoid for prophylaxis in G4–5 | Give after HD; dialysed partially | Bactrim (Roche), generic |
| Metronidazole oral/IV | 500 mg q8h | No adjustment (<7 days) | 500 mg q12h for prolonged courses; metabolite accumulates | No supplement needed (not removed by HD) | Flagyl (Sanofi), generic |
| Azithromycin oral | 500 mg OD × 3–5 days | No adjustment | No adjustment (biliary) | No supplement | Zithromax (Pfizer), generic |
| Clarithromycin oral CYP3A4 inhibitor | 500 mg q12h | 250 mg q12h | 250 mg q12h (max 500 mg/day if CrCl <30) | No supplement | Klacid (Abbott), generic |
| Fluconazole oral/IV | 100–400 mg q24h | 50% dose reduction if CrCl <50 | 50% dose reduction; full loading dose | Full dose after each HD session (significantly dialysed) | Diflucan (Pfizer), generic |
| Nitrofurantoin Avoid CKD | 100 mg q12h | AVOID if CrCl <30 — inadequate urine drug concentration + peripheral neuropathy risk. Use cephalosporins or quinolones instead for UTI in CKD. | — | ||
Aminoglycosides not listed — avoid in non-dialysis CKD. In dialysis: 1.5 mg/kg gentamicin post-HD with peak/trough TDM only; seek ID or nephrology guidance.
High-Risk Pitfalls — What Catches Clinicians Out
If a CKD patient on cefepime becomes confused or develops myoclonus: assume cefepime toxicity first. Hold drug, check EEG, obtain nephrology review. Recovery is usually complete after dose reduction or cessation. Dose-reduce aggressively (see table) — 500 mg q24h in dialysis is often sufficient for susceptible organisms.
Substitute: cephalexin 500 mg q12h × 3–5 days; or cefuroxime 250 mg q12h; or levofloxacin 250 mg q24h × 3 days (check local resistance pattern first).
Trimethoprim also blocks tubular creatinine secretion → serum creatinine rises 0.1–0.3 mg/dL without any change in true GFR. This frequently triggers unnecessary nephrology referrals or RAASi dose reduction.
Rules: avoid TMP-SMX for prophylaxis in CKD G4–5 (K⁺ risk); if treating a UTI in G3, check K⁺ at day 3–5; if K⁺ rises >0.5 mEq/L above baseline, switch antibiotic.
Affected drugs: tacrolimus, cyclosporine, finerenone, simvastatin/atorvastatin (myopathy risk), colchicine (toxicity risk), DOACs (rivaroxaban, apixaban levels rise).
Preferred alternative: azithromycin — equally effective for respiratory infections, no relevant CYP3A4 interaction.
If aminoglycoside is truly necessary (e.g., gentamicin for synergy in endocarditis, ID-directed): single daily dosing (extended interval), peak (30 min post-dose) and trough (pre-next-dose) monitoring, and limit to the shortest possible course. In dialysis patients, give post-HD only with full TDM.
Avoid triple nephrotoxin combination: aminoglycoside + vancomycin + loop diuretic — this combination produces AKI in >30% of patients even with normal baseline GFR.
In HD patients: give full loading dose (25–30 mg/kg for serious infections); supplement 500 mg–1 g when pre-HD trough <15 mg/L (typically every 3–7 days depending on dialysis membrane). High-flux and high-efficiency membranes remove significantly more vancomycin.
Access Infections in HD — Empiric Antibiotic Selection
AV fistula / graft exit-site infection or bacteraemia
Empiric (pending blood cultures):
MSSA likely: Cefazolin 1 g IV after each HD session
MRSA suspected (prior MRSA, nasal carrier, healthcare exposure): Vancomycin per TDM dosing above.
De-escalate to cefazolin once culture confirms MSSA — do not continue vancomycin for susceptible Staph aureus (inferior outcomes + nephrotoxicity).
Duration: 6 weeks for confirmed bacteraemia (rule out endocarditis with echo if Staph aureus); 2 weeks for exit-site-only without bacteraemia.
Tunnelled catheter (TCC) infection
Exit site only (no fever, no bacteraemia): topical mupirocin + systemic antibiotic for 2 weeks; can attempt catheter salvage.
Tunnel infection or bacteraemia: remove catheter — do not attempt to salvage. Empiric vancomycin; replace catheter only after 2–3 negative blood cultures and apyrexial ≥48 h.
Staph aureus bacteraemia with TCC: mandatory catheter removal + minimum 4 weeks IV antibiotics + rule out endocarditis (TEE preferred). Oral step-down is not appropriate for Staph aureus bacteraemia in this population.
PD Peritonitis — ISPD 2022 Empiric Protocol
Add vancomycin 15–30 mg/kg IP if prior MRSA, recent hospitalisation, or severe presentation.
Gram-negative cover: Ceftazidime 15 mg/kg IP in one daily long-dwell exchange. Alternative: gentamicin 0.6 mg/kg IP (single daily long-dwell) if ceftazidime unavailable — monitor residual renal function.
Duration: 14 days for most organisms (21 days for Staph aureus, Enterococcus, Pseudomonas, fungal — latter requires catheter removal + antifungal).
Quick Reference — Antibiotic Stewardship in CKD
- Use CrCl (Cockcroft-Gault) — not eGFR — for dose adjustment
- Loading dose: full standard dose regardless of CKD stage
- Cefepime: dose-reduce aggressively; confusion + myoclonus = stop drug immediately
- Nitrofurantoin: avoid if CrCl <30 — ineffective AND toxic; use cephalexin/levofloxacin
- TMP-SMX: raises K⁺ and creatinine; check K⁺ at day 3–5; avoid prophylaxis in G4–5
- Clarithromycin: CYP3A4 inhibitor — doubles tacrolimus/finerenone levels; use azithromycin instead
- Ceftriaxone + moxifloxacin + azithromycin: no renal dose adjustment
- Aminoglycosides: avoid in non-dialysis CKD; post-HD only in dialysis with TDM
- Never combine aminoglycoside + vancomycin + loop diuretic (triple nephrotoxin)
- HD access bacteraemia: cefazolin 1 g post-HD for MSSA; vancomycin TDM for MRSA
- Vancomycin HD: supplement post-session; AUC-guided preferred over trough-only
- PD peritonitis: IP cefazolin + IP ceftazidime empirically; remove catheter for fungal/refractory
Anti-Tuberculosis Medications in CKD and ESKD
 Anti-TB Drugs in CKD — RIPE dose adjustments by CrCl and post-dialysis dosing rules.
Anti-TB Drugs in CKD — RIPE dose adjustments by CrCl and post-dialysis dosing rules.
First-Line RIPE Agents — Dose Adjustment by CKD Stage
| Drug | Normal dose | CrCl 30–59 | CrCl 10–29 / G5 | HD (thrice-weekly) | PD | Key toxicity |
|---|---|---|---|---|---|---|
| Isoniazid (H) Hepatic acetylation |
5 mg/kg/day (max 300 mg) | No change | No change | No change; give after HD session (dialyzable) |
No change | Hepatotoxicity; peripheral neuropathy — give pyridoxine 25–50 mg/day in all CKD patients |
| Rifampicin (R) Biliary elimination |
10 mg/kg/day (max 600 mg) | No change | No change | No change; not significantly dialyzed | No change | Hepatotoxicity; potent CYP inducer — reduces levels of tacrolimus, ciclosporin, DOACs, azole antifungals; orange discoloration of body fluids |
| Pyrazinamide (Z) Hepatic; renal metabolite excretion |
25–30 mg/kg 3×/week | 25 mg/kg 3×/week | 25 mg/kg 3×/week | 25–35 mg/kg 3×/week — give after HD (metabolite dialyzable) |
12–20 mg/kg/day | Hyperuricemia (accentuated in CKD); hepatotoxicity; arthralgia. Monitor uric acid; add allopurinol if symptomatic gout (dose-adjust to CrCl) |
| Ethambutol (E) ~80% renal excretion |
15–25 mg/kg/day | 15 mg/kg/day every 24–36 h | 15 mg/kg every 48 h | 15–25 mg/kg 3×/week — give after HD (60% dialyzable) |
15 mg/kg every 24–48 h | Optic neuritis (dose-dependent, accentuated by accumulation in renal failure) — monthly visual acuity + red-green color testing mandatory |
Second-Line Agents (Drug-Resistant TB) — CKD Considerations
| Drug | Normal | CrCl 30–59 | CrCl <30 / G5 | HD | Notes |
|---|---|---|---|---|---|
| Levofloxacin | 750–1000 mg/day | 750 mg/day | 750–1000 mg every 48 h | 750–1000 mg 3×/week after HD | QTc prolongation; tendinopathy; seizures (accumulation). Preferred FQ for TB-CKD over moxifloxacin |
| Moxifloxacin | 400 mg/day | No change | No change | No change; not significantly dialyzed | QTc prolongation; no dose adjustment but avoid with other QT-prolonging drugs |
| Amikacin / Streptomycin Aminoglycosides |
15 mg/kg/day | Extend to 24–48 h; TDM mandatory | Avoid if possible; if unavoidable: 12–15 mg/kg 2–3×/week with TDM | Give after HD; TDM pre- and post-HD; target Cmax 20–35, Cmin <5 μg/mL | Nephrotoxic + ototoxic — cumulative, irreversible. Reserve for MDR-TB only in CKD. |
| Cycloserine | 250–500 mg twice daily | 250 mg twice daily | 250 mg/day | 250 mg/day; supplement pyridoxine 50–100 mg/day | CNS toxicity (seizures, psychosis) accentuated by accumulation. TDM recommended (Cmax 20–35 μg/mL) |
| Bedaquiline | 400 mg/day ×2 wk → 200 mg 3×/week ×22 wk | No change | Limited data; no formal adjustment — monitor QTc | Not significantly dialyzed; monitor QTc | QTc prolongation (synergistic with FQs, clofazimine); hepatotoxicity; long half-life (~5 months) |
| Linezolid | 600 mg twice daily or 600 mg/day | No change | No change (hepatic); metabolite accumulates — monitor CBC | Give after HD; supplement B6 | Myelosuppression (thrombocytopenia > anemia — accentuated by uremia); serotonin syndrome; optic neuritis with prolonged use |
| Clofazimine | 100 mg/day | No change | No change | No change; not dialyzed | QTc prolongation; skin discoloration (reddish-brown); GI intolerance; very long half-life |
| Para-aminosalicylic acid (PAS) | 8–12 g/day | Caution | Avoid — metabolite accumulates; sodium load worsens HTN/fluid retention | Avoid | Largely replaced by newer agents in MDR-TB regimens |
High-Risk Pitfalls in TB + CKD
Monitoring Schedule During TB Treatment in CKD
| Parameter | Frequency | Rationale |
|---|---|---|
| LFTs (AST, ALT, bilirubin) | Baseline, then monthly | All first-line agents are hepatotoxic; uremia blunts early symptoms |
| Visual acuity + red-green color vision | Baseline, then monthly (if on EMB) | Ethambutol optic neuritis — irreversible if delayed |
| Serum uric acid | Baseline, monthly (if on PZA) | Pyrazinamide + CKD = compounding hyperuricemia |
| CBC | Baseline, monthly (if on linezolid) | Thrombocytopenia and anemia — accentuated in uremia |
| ECG (QTc) | Baseline, 2 wk, 4 wk, then monthly (if on BDQ/CFZ/FQ) | QTc prolongation risk, especially with stacked regimens |
| Audiometry | Baseline + monthly (if on aminoglycosides) | Irreversible ototoxicity from accumulation |
| Tacrolimus / ciclosporin trough | Daily × 1–2 weeks when rifampicin started or stopped | CYP3A4 induction/deinduction causes large unpredictable level swings |
| Neuropathy assessment | Monthly (if on INH, cycloserine, linezolid) | Peripheral neuropathy — give pyridoxine 25–50 mg/day throughout |
Quick Reference — Anti-TB in CKD/ESKD
- Isoniazid & rifampicin: no dose adjustment in any CKD stage
- Pyrazinamide: 3×/week dosing in CrCl <30 and HD; give post-HD
- Ethambutol: every 48 h in CrCl <30; 3×/week post-HD
- All dialyzable agents: dose immediately after HD session
- Pyridoxine 25–50 mg/day: mandatory with INH in every CKD patient
- Rifampicin + tacrolimus: expect 10–15× level drop — daily TDM when starting/stopping
- Ethambutol: monthly visual acuity + color vision — stop at first symptom
- PZA: monitor uric acid; add allopurinol if symptomatic gout develops
- Aminoglycosides: reserve for MDR-TB only; TDM + audiometry mandatory
- Levofloxacin preferred over moxifloxacin for FQ-based regimens in CKD
- QTc stack (BDQ + CFZ + FQ): ECG baseline, 2 wk, 4 wk, monthly
- Cycloserine accumulates in ESKD: reduce to 250 mg/day; watch for seizures/psychosis
HIV Antiretroviral Medications in CKD and ESKD
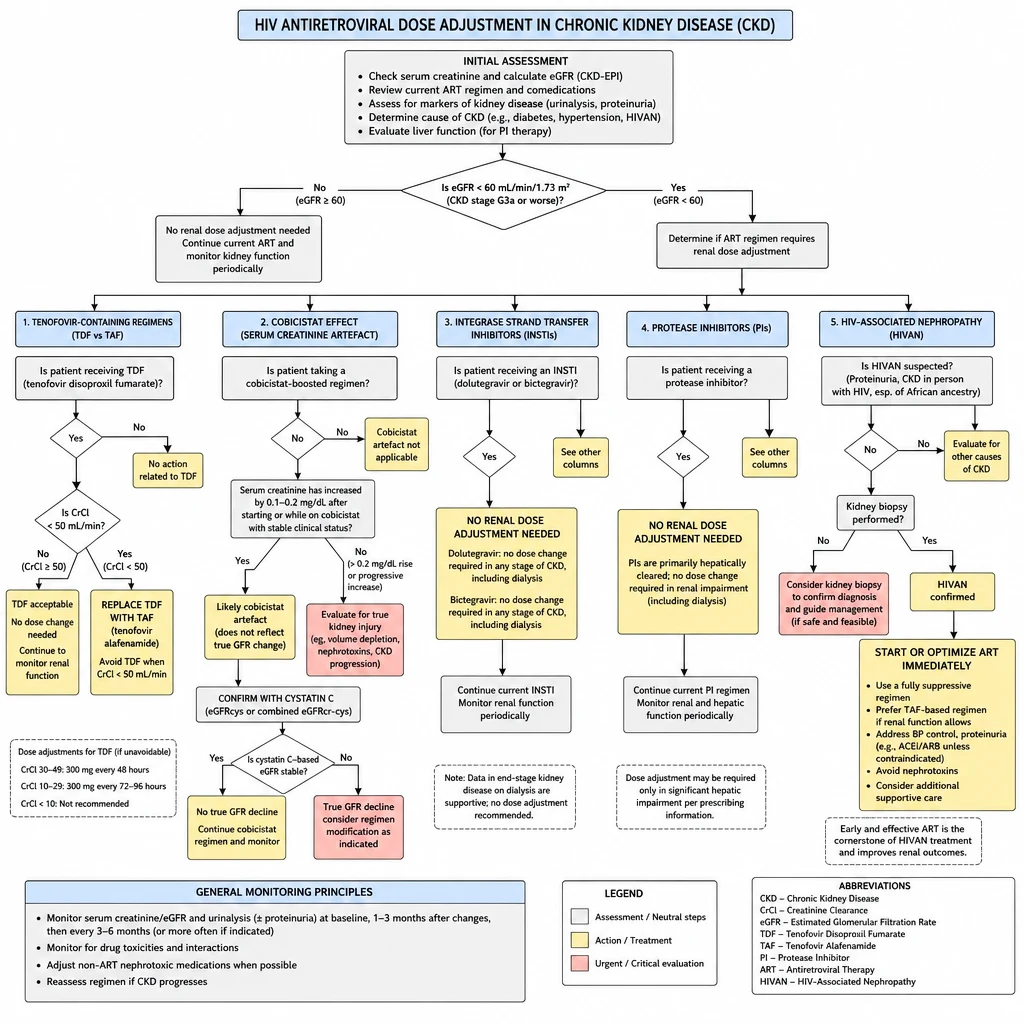 HIV ARV in CKD — TDF nephrotoxicity, cobicistat artifact, and integrase inhibitor preference.
HIV ARV in CKD — TDF nephrotoxicity, cobicistat artifact, and integrase inhibitor preference.
NRTI Backbone — Dose Adjustment by Renal Function
| Drug | CrCl ≥50 | CrCl 30–49 | CrCl 10–29 | HD | PD | Key renal concern |
|---|---|---|---|---|---|---|
| Tenofovir DF (TDF) Viread; in Truvada, Atripla |
300 mg q24h | 300 mg q48h | 300 mg q72–96h (limited data — avoid if possible) | 300 mg after each HD session (~weekly) (dialyzable) |
Avoid — inadequate clearance data; high nephrotoxicity risk | Proximal tubular toxicity (Fanconi syndrome), AKI, CKD progression. Prefer TAF in CrCl <50. |
| Tenofovir alafenamide (TAF) In Descovy, Genvoya, Symtuza, Biktarvy |
25 mg q24h (or 10 mg with booster) | No change | No change | No change; not significantly dialyzed | No change | Preferred TDF alternative in CrCl <50. ~90% lower plasma tenofovir levels than TDF — less tubular toxicity. Limited data in CrCl <15. |
| Emtricitabine (FTC) In Truvada, Descovy, Biktarvy |
200 mg q24h | 200 mg q48h | 200 mg q72h | 200 mg after each HD (3×/week) (dialyzable ~30%) |
200 mg q96h | Renally excreted — dose-extend in CKD. Rarely causes toxicity at adjusted doses. |
| Lamivudine (3TC) Epivir; in Kivexa, Triumeq |
300 mg q24h or 150 mg q12h | 150 mg q24h | 100 mg loading → 50 mg q24h | 50 mg after HD (3×/week) (dialyzable) |
25–50 mg q24h | Renally excreted; well-tolerated at adjusted doses. Preferred NRTI partner in low-resource HD settings. |
| Abacavir (ABC) In Kivexa, Triumeq |
600 mg q24h or 300 mg q12h | No change | No change | No change; not dialyzed (hepatic glucuronidation) | No change | Hepatically metabolized — no renal dose adjustment. HLA-B*57:01 screen mandatory before use (hypersensitivity). Cardiovascular risk with prolonged use. |
| Zidovudine (AZT) Retrovir; second-line only |
300 mg q12h | No change | 100 mg q8h (metabolite accumulation) | 100 mg q6–8h; supplement after HD | 100 mg q8h | Myelosuppression (anemia, neutropenia) — accentuated by CKD-related anemia and EPO deficiency. Use only when no alternative available. |
INSTIs, NNRTIs, and Boosted PIs — Renal Adjustment Summary
| Class / Drug | Renal adjustment needed? | HD | Notes |
|---|---|---|---|
| Dolutegravir (DTG) In Triumeq, Dovato |
No — hepatic UGT1A1/CYP3A | No change; not dialyzed | Preferred INSTI backbone in CKD/ESKD. Raises serum creatinine ~0.1 mg/dL via OCT2 inhibition (artifact, not true GFR fall). Safe in all stages. |
| Bictegravir (BIC) In Biktarvy |
No — hepatic CYP3A/UGT1A1 | No change; not dialyzed | Limited data in CrCl <30 and HD. Generally considered safe; part of preferred Biktarvy (BIC/TAF/FTC) regimen in stable CKD ≥15. |
| Raltegravir (RAL) Isentress |
No — hepatic glucuronidation | No change; not dialyzed | Well-characterized in HD — older INSTI, used when newer options unavailable. No creatinine artifact. |
| Elvitegravir/cobicistat In Stribild, Genvoya |
Avoid if CrCl <70 (Stribild) or <30 (Genvoya/TAF) | Not recommended — cobicistat unpredictable in HD | Cobicistat raises creatinine ~0.1–0.2 mg/dL (artifact). Stribild contains TDF — avoid in CrCl <70. Genvoya (TAF-based) avoid in CrCl <30. |
| Efavirenz (EFV) In Atripla; second-line |
No — hepatic CYP2B6 | No change; not dialyzed | Neuropsychiatric side effects (vivid dreams, depression) — CKD patients with uremia may be more susceptible. Atripla contains TDF; substitute individually if CrCl <50. |
| Rilpivirine (RPV) In Odefsey, Complera |
No — hepatic CYP3A4 | No change; not dialyzed | Odefsey (RPV/TAF/FTC) is an option in CKD — check that FTC dose is adjusted. Requires acid environment for absorption — avoid PPIs. |
| Darunavir/ritonavir (DRV/r) Prezista + Norvir |
No — hepatic CYP3A4 | No change; not dialyzed | Ritonavir raises creatinine slightly via OCT2 inhibition (artifact). Monitor LFTs — both are hepatotoxic. Preferred PI in CKD when INSTI not available. |
| Darunavir/cobicistat (DRV/c) Rezolsta, Symtuza |
Avoid in CrCl <70 (Symtuza has TDF); DRV/c alone: avoid <30 | Not recommended | Same cobicistat creatinine artifact concern. Symtuza (DRV/c/TAF/FTC) is an option in CrCl ≥15 if TAF-based, but limited HD data. |
| Lopinavir/ritonavir (LPV/r) Kaletra; second-line |
No — hepatic | No change; not dialyzed | GI intolerance, dyslipidemia, and hepatotoxicity — generally replaced by INSTIs. Retained as second-line option in resource-limited settings. |
Preferred Regimens by CKD Stage
Critical Drug Interactions in HIV + CKD
HIV-Associated Nephropathy and TDF Nephrotoxicity
| HIVAN | TDF Nephrotoxicity | |
|---|---|---|
| Mechanism | Direct HIV infection of podocytes and tubular cells; collapsing FSGS pattern | Mitochondrial toxicity in proximal tubular cells → Fanconi syndrome |
| Presentation | Heavy proteinuria, rapid CKD progression, echogenic kidneys on US | Normoglycemic glucosuria, hypophosphatemia, low uric acid, tubular proteinuria (β2-microglobulin), AKI on CKD |
| Diagnosis | Kidney biopsy (collapsing FSGS); most common in African ancestry | Urine phosphate reabsorption <80%, urine β2-microglobulin, CrCl trend since TDF start |
| Management | Start/optimize ART (INSTIs preferred); ACEi/ARB for proteinuria; corticosteroids controversial | Stop TDF; switch to TAF or ABC-based regimen; renal recovery partial (months); replace phosphate and electrolytes |
Renal Monitoring in PLHIV on ART
| Parameter | Frequency | Rationale |
|---|---|---|
| Serum creatinine + CrCl (C-G) | Baseline, then every 3–6 months on TDF; every 6–12 months on TAF/ABC | TDF nephrotoxicity detection; dose-adjustment thresholds |
| Urine dipstick (protein, glucose) | Baseline, then annually; more frequent on TDF | HIVAN (proteinuria) and Fanconi syndrome (glucosuria) screening |
| Serum phosphate | Baseline, then every 6 months on TDF | Proximal tubular phosphate wasting — early TDF toxicity marker |
| Urine β2-microglobulin or RBP | If TDF + CrCl fall or phosphaturia detected | Confirms proximal tubular dysfunction before Fanconi syndrome is overt |
| Bone density (DXA) | Baseline in CKD G3+; every 2 years | TDF and CKD both cause bone loss; TAF has less bone effect |
| Tacrolimus / ciclosporin trough | Daily × 2 weeks when boosted ARV started or stopped (transplant patients) | Cobicistat/ritonavir CYP3A4 inhibition causes rapid CNI level surge |
| HIV viral load + CD4 | Every 3–6 months (or per HIV program protocol) | Virologic failure increases HIVAN and opportunistic infection risk |
Quick Reference — HIV ARVs in CKD/ESKD
- Switch TDF → TAF when CrCl falls below 50 mL/min
- Biktarvy (BIC/TAF/FTC) or DTG + 3TC: preferred in CKD G3b–G5
- ABC: no renal adjustment; requires HLA-B*57:01 screening first
- 3TC and FTC: dose-extend in CrCl <50; give post-HD on dialysis days
- Dolutegravir and raltegravir: no renal adjustment; safe at all CKD stages
- Cobicistat creatinine rise (0.1–0.2 mg/dL): artifact — confirm with cystatin C before switching ARVs
- Rifampicin + DTG: double DTG to 50 mg twice daily; BIC contraindicated with rifampicin
- Rifampicin + boosted PI: contraindicated — substitute rifabutin
- Cobicistat/ritonavir + tacrolimus (post-transplant): 10–100× CNI level rise — prefer unboosted INSTI
- NSAIDs + TDF: avoid — synergistic nephrotoxicity
- PPIs + rilpivirine or atazanavir: avoid — absorption failure
- HIVAN: start/optimize ART early; ACEi/ARB for proteinuria; biopsy if diagnosis uncertain
Stone Dissolution and Medical Expulsion Therapy
Stone type determines treatment — uric acid and cystine stones dissolve with alkalinization; calcium stones do not. Medical expulsion therapy (MET) with alpha-blockers accelerates passage of stones ≤10 mm and is first-line before urologic referral.
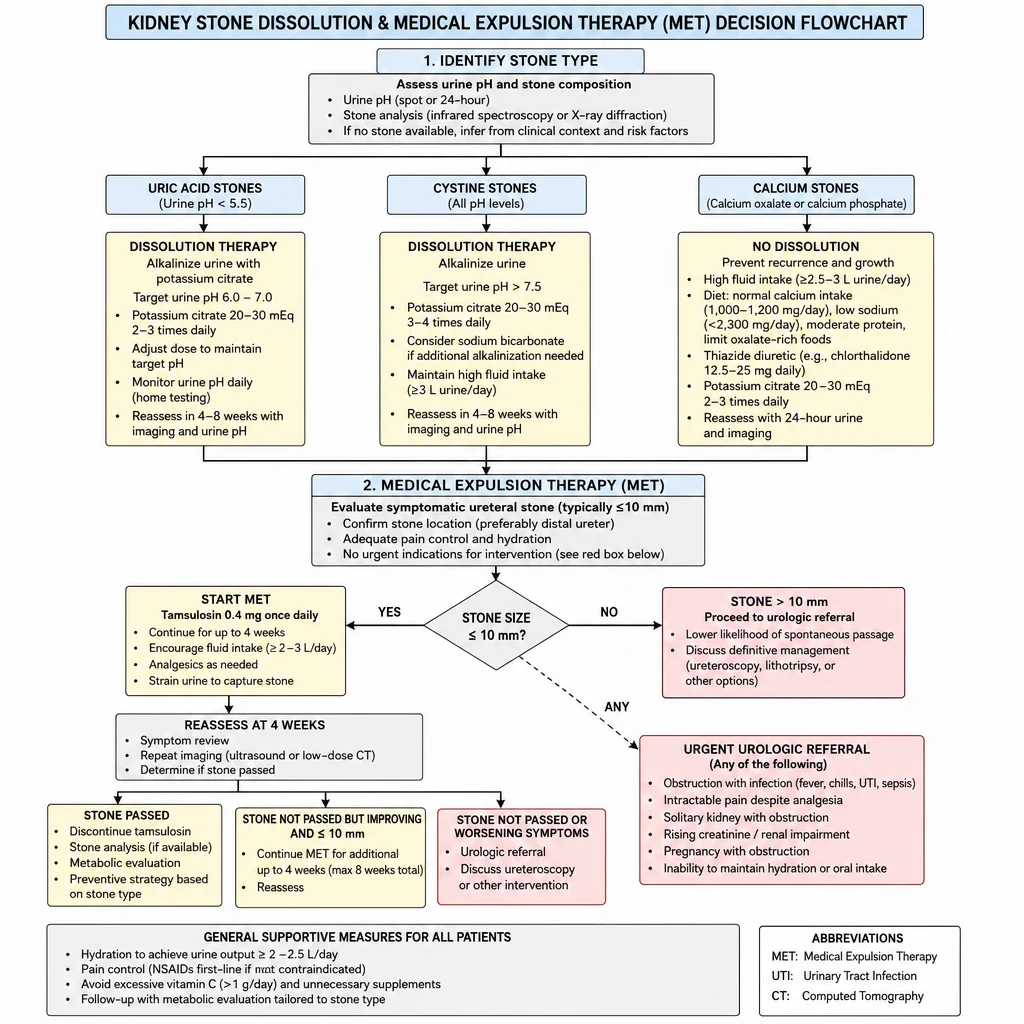 Stone Dissolution & MET — uric acid alkalinization, alpha-blocker expulsion therapy, and urologic referral criteria.
Stone Dissolution & MET — uric acid alkalinization, alpha-blocker expulsion therapy, and urologic referral criteria.
Medical Expulsion Therapy (MET) — Stones ≤10 mm
| Drug | Dose | Local brands | Evidence / Notes |
|---|---|---|---|
| Tamsulosin (alpha-1 blocker) First-line MET |
0.4 mg once daily after meals × 4 weeks (or until passage) | Harnal OCAS (Astellas) ~₱55–75/cap Contiflo OD, generic ~₱20–35/cap |
Relaxes ureteral smooth muscle — increases passage rate and reduces time to passage. Best evidence for distal ureteral stones 5–10 mm. Continue ≤4 weeks; refer if no passage. |
| Silodosin (alpha-1A blocker) | 8 mg once daily with meals × 4 weeks | Urorec (Recordati) ~₱80–100/cap | More uroselective than tamsulosin; similar efficacy for distal ureteral stones. Retrograde ejaculation more common. Alternative if tamsulosin not tolerated. |
| Nifedipine XL (CCB) Second-line MET |
30 mg once daily × 4 weeks | Adalat OROS (Bayer) ~₱20–30/tab Generic nifedipine XL |
Relaxes ureteral smooth muscle via calcium-channel blockade. Inferior to tamsulosin in meta-analyses but still used when alpha-blockers are contraindicated (e.g., severe orthostatic hypotension). May help concurrent HTN. |
Urinary Alkalinization and Stone Dissolution Agents
| Drug | Mechanism | Dose | Local brand / cost | Target urine pH | Stone types treated | Key cautions |
|---|---|---|---|---|---|---|
| Potassium citrate First-line alkalinizer |
Alkalinizes urine; citrate chelates Ca²⁺ reducing calcium supersaturation; inhibits crystal growth | 10–20 mEq (1–2 tabs) TID with meals or after meals; max 100 mEq/day | Urocit-K (Mission) ~₱25–40/tab Potassium citrate generic ~₱15–25/tab |
6.5–7.0 (uric acid) 6.0–6.5 (calcium oxalate prevention) |
Uric acid stones (dissolution + prevention); calcium oxalate (prevention); hypocitraturic calcium stones; distal RTA | Hyperkalemia risk — check K⁺ at baseline, 1 month, then q3–6 months. Avoid in CKD G4–5 unless K⁺ closely monitored. Take with meals — GI irritation if taken fasting. |
| Sodium bicarbonate | Systemic alkalinization → urine pH rise; cheap and widely available | 650 mg–1 g TID–QID (titrate to urine pH); or ½–1 tsp baking soda in water TID | Generic NaHCO₃ tabs ~₱2–5/tab Baking soda (pharmacy-grade) ~₱50/box |
6.5–7.0 (uric acid) >7.5 (cystine) |
Uric acid stones; cystine stones (adjunct); metabolic acidosis in CKD | Sodium load — worsens fluid retention, HTN, and edema. Avoid in CHF, severe HTN, anasarca. Prefer potassium citrate in hypertensive or volume-sensitive patients. Alkalosis risk with overuse. |
| Ural (Sodium Citrotartrate) | Urinary alkalinizer; citrate + tartrate combination → raises urine pH | 1 sachet (4 g) dissolved in water, 3–4× daily; continue 2–4 weeks for acute uric acid stone dissolution | Ural sachets (Alphapharm/Zuellig) ~₱18–25/sachet Available OTC in Mercury, Watsons |
6.5–7.0 | Uric acid stones (acute dissolution); symptomatic UTI alkalinization; dysuria relief | Contains sodium — same cautions as sodium bicarbonate for fluid-sensitive patients. Not a potassium source — does not correct hypocitraturia as effectively as potassium citrate long-term. Good for short-course acute dissolution. |
| Sambong (Blumea balsamifera) DOH-approved Philippine herbal medicine |
Diuretic and anti-urolithiasis properties; proposed mechanism: increased urine output + possible inhibition of crystal aggregation | 500 mg–1 g TID (standardized extract); take with adequate fluid (≥2 L/day water) | Sambong tablet (various local manufacturers) ~₱5–15/tab Available in Mercury, Rose Pharmacy, generic brands |
No direct pH effect — mechanism is diuretic + crystal inhibition | Small renal stones (<5 mm); stone passage adjunct; stone prevention; adjunct to MET | DOH-PITAHC approved for urolithiasis. Evidence base is limited to smaller local RCTs — inform patients this is herbal/adjunct therapy, not a replacement for potassium citrate or urologic evaluation. Adequate hydration is essential for effect. Safe in most patients; limited data in severe CKD. |
| Allopurinol | Xanthine oxidase inhibitor → reduces uric acid production → lower urinary uric acid | CrCl ≥60: 300 mg/day; CrCl 30–59: 100–200 mg/day; CrCl <30: 100 mg/day or every other day; HD: 100 mg after each HD session | Zyloric (Aspen) ~₱8–15/tab Generic allopurinol ~₱3–8/tab |
No direct pH effect | Uric acid stones (hyperuricosuria); calcium oxalate stones with hyperuricosuria (reduces nucleation); gout with urolithiasis | Start low, titrate slowly — allopurinol hypersensitivity (SCAR, SJS) risk especially in Han Chinese: HLA-B*58:01 screen recommended before starting. Rash → stop immediately. Dose-reduce in CKD (see table). Interactions: azathioprine, 6-MP (toxic accumulation — reduce AZA/6-MP dose by 75%). |
| Hydrochlorothiazide (HCTZ) | Reduces urinary calcium excretion by enhancing proximal tubular Ca²⁺ reabsorption | 25 mg once or twice daily (hypercalciuria); ensure adequate dietary sodium restriction for maximal effect | Generic HCTZ ~₱2–5/tab; also in combination products | No pH effect | Calcium oxalate stones with hypercalciuria; recurrent calcium stones | Hypokalemia common — supplement K⁺ or combine with potassium citrate (addresses both hypokalemia AND hypocitraturia from K⁺ loss). Monitor electrolytes, uric acid (HCTZ raises uric acid). Avoid in severe CKD (CrCl <30) — ineffective and hyperuricemia risk. |
| D-penicillamine / Tiopronin | Thiol-disulfide exchange → forms soluble cysteine-drug complex; reduces free cystine in urine | D-penicillamine: 250 mg QID (limited local supply); Tiopronin (Thiola): 800–1200 mg/day in divided doses |
Limited local availability — may require compassionate supply or referral to tertiary center | No pH effect (used with alkalinization) | Cystinuria — severe or recurrent cystine stones not controlled by alkalinization alone | Significant toxicity: nephrotoxicity, proteinuria, rash, bone marrow suppression, autoimmune reactions. Reserve for cystinuria refractory to hydration + alkalinization + dietary modification. Specialist referral required. |
Stone-Type Specific Treatment Protocols
Prevention: Potassium citrate 10–20 mEq TID long-term + allopurinol 100–300 mg/day if hyperuricosuria or recurrent stones. Increase fluid intake to maintain urine output ≥2.5 L/day. Low-purine diet.
Hypocitraturia: Potassium citrate 20–30 mEq TID — citrate is the single most effective inhibitor of calcium oxalate crystallization.
Hyperoxaluria: Low-oxalate diet (avoid spinach, nuts, chocolate, beetroot); calcium supplementation WITH meals (not fasting) to bind gut oxalate.
Fluid intake ≥2.5 L/day remains the single most impactful intervention across all calcium stone subtypes.
Monitoring During Stone Prevention Therapy
| Drug | Baseline | Ongoing | Watch for |
|---|---|---|---|
| Potassium citrate | Serum K⁺, creatinine, urine pH | K⁺ at 1 month, then q3–6 months; urine pH dipstick weekly (patient self-monitors) | Hyperkalemia (esp. CKD G3+); GI intolerance |
| Sodium bicarbonate / Ural | BP, electrolytes, weight | BP and weight monthly; electrolytes q3 months | HTN, fluid retention, metabolic alkalosis |
| Allopurinol | Serum uric acid, CrCl, LFTs, FBC; HLA-B*58:01 screen in Han Chinese | Uric acid q3–6 months; LFTs + FBC at 3 months then annually | Rash (→ stop immediately; Stevens-Johnson risk); azathioprine toxicity |
| HCTZ | K⁺, Na⁺, uric acid, creatinine | Electrolytes at 4 weeks, then q3–6 months; uric acid annually | Hypokalemia, hyponatremia, hyperuricemia (may precipitate gout) |
| Tamsulosin (MET) | BP (orthostatic) | Stone passage — KUB or CT at 4 weeks if no passage | Orthostatic hypotension, retrograde ejaculation |
Quick Reference — Stone Dissolution and MET
- MET first-line: tamsulosin 0.4 mg/day (Harnal) × 4 weeks for ureteral stones ≤10 mm
- Uric acid stones: alkalinize to urine pH 6.5–7.0 — stones dissolve; CT confirms
- Potassium citrate (Urocit-K): preferred alkalinizer — addresses both pH and citrate deficiency
- Ural sachets: good for short-course acute dissolution; OTC, affordable, easy to use
- Sodium bicarb: cheap alternative but avoid in HTN, CHF, edema — sodium load
- Sambong (Blumea balsamifera): DOH-approved herbal adjunct; ≥2 L/day fluid essential
- Calcium oxalate stones: cannot dissolve — prevent with potassium citrate + fluid + diet
- Allopurinol: dose-adjust in CKD; HLA-B*58:01 screen before use in Han Chinese patients
- HCTZ for hypercalciuria: always pair with potassium citrate to prevent hypokalemia
- Struvite: complete stone removal required; medical therapy alone insufficient
- Fluid intake ≥2.5 L/day: single most effective intervention for all stone types
- 24-hour urine collection: guides targeted prevention — order after first recurrence

W. G. M. Rivero, MD, FPCP, DPSN
Nephrologist · Internal Medicine · Philippines. These protocols synthesize KDIGO 2024, ACC/AHA guidance, and the practical realities of managing CKD and related conditions in the Philippine outpatient and inpatient setting. Always individualize to your patient.
FPCP · Diplomate, Philippine Society of Nephrology